241989
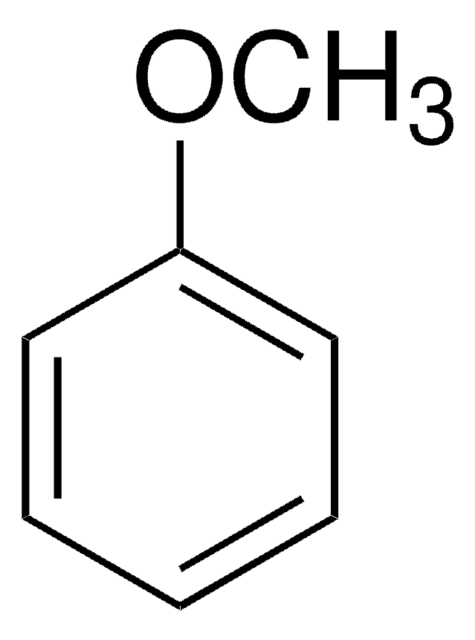
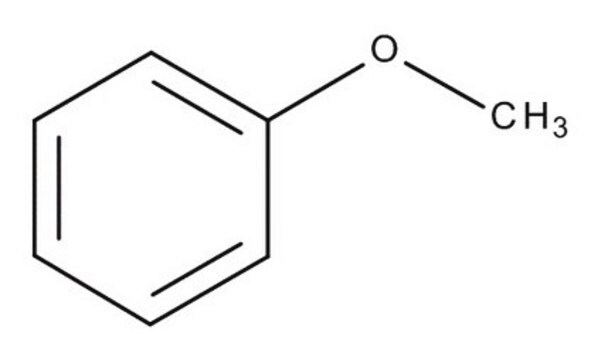
Ethoxybenzene
99%
Synonym(s):
Phenetole, Ethoxybenzene, Ethyl phenyl ether
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
C6H5OC2H5
CAS Number:
Molecular Weight:
122.16
Beilstein:
636270
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
liquid
refractive index
n20/D 1.507 (lit.)
bp
169-170 °C (lit.)
mp
−30 °C (lit.)
solubility
alcohol: freely soluble(lit.)
diethyl ether: freely soluble(lit.)
water: insoluble(lit.)
density
0.966 g/mL at 25 °C (lit.)
functional group
phenoxy
SMILES string
CCOc1ccccc1
InChI
1S/C8H10O/c1-2-9-8-6-4-3-5-7-8/h3-7H,2H2,1H3
InChI key
DLRJIFUOBPOJNS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
Ethoxybenzene (Phenetole) was used as an analyte in assaying the performance of the porous graphitic carbon (PGC) particles.
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
H Ohi et al.
Xenobiotica; the fate of foreign compounds in biological systems, 22(11), 1329-1337 (1992-11-01)
1. The effects of oxygen concentration were studied on the metabolic pathways of anisole homologues (anisole, phenetole and isopropoxybenzene) catalysed by liver microsomes from phenobarbital-treated rats. 2. With increase of oxygen concentration, the rate of anisole o-hydroxylation reached a plateau
Wojciech Piatkowski et al.
Journal of chromatography. A, 1003(1-2), 73-89 (2003-08-06)
The competitive adsorption behavior of the binary mixture of phenetole (ethoxy-benzene) and propyl benzoate in a reversed-phase system was investigated. The adsorption equilibrium data of the single-component systems were acquired by frontal analysis. The same data for binary mixtures were
Niklas Helle et al.
European journal of mass spectrometry (Chichester, England), 25(1), 142-156 (2019-02-19)
The vibronic structure of the first electronically excited state S1 and ionic ground state D0 of phenetole has been investigated by means of resonance enhanced multi photon ionization (REMPI) and mass analyzed threshold ionization (MATI) spectroscopy. The vibronic levels were
David S Jensen et al.
Journal of chromatography. A, 1218(46), 8362-8369 (2011-10-19)
Porous graphitic carbon (PGC) particles were functionalized/passivated in situ in packed beds at elevated temperature with neat di-tert-amylperoxide (DTAP) in a column oven. The performance of these particles for high performance liquid chromatography (HPLC) was assayed before and after this
Isamu Shiina et al.
Bioorganic & medicinal chemistry, 15(24), 7599-7617 (2007-10-02)
Two new synthetic pathways to the anti-cancer agent tamoxifen and its derivatives were developed. The first route involved the aldol reaction of benzyl phenyl ketone with acetaldehyde followed by Friedel-Crafts substitution with anisole in the presence of Cl(2)Si(OTf)(2) to produce
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service