All Photos(1)
About This Item
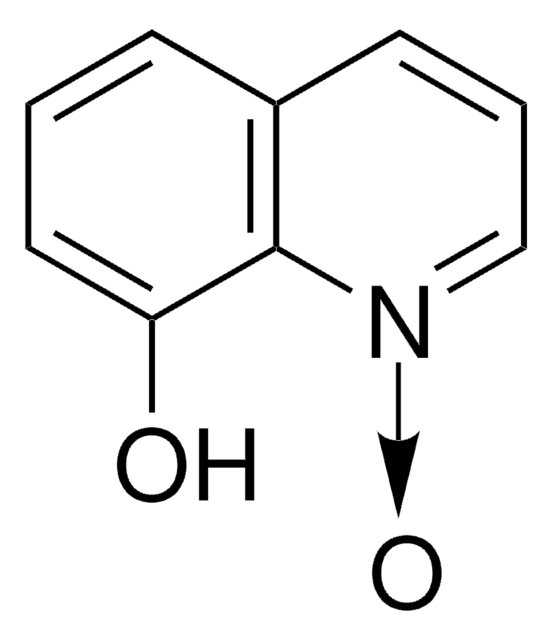
Empirical Formula (Hill Notation):
C9H6N2O2
CAS Number:
Molecular Weight:
174.16
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
mp
89-91 °C (lit.)
functional group
nitro
SMILES string
[O-][N+](=O)c1cccc2cccnc12
InChI
1S/C9H6N2O2/c12-11(13)8-5-1-3-7-4-2-6-10-9(7)8/h1-6H
InChI key
OQHHSGRZCKGLCY-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
8-Nitroquinoline was used to prepare furazano [3,4-h] quinoline. It was also used to synthesize corresponding 2-substituted phenoxy-6-methoxy-8-aminoquinoline.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Carc. 2 - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
M Hasegawa et al.
Yakugaku zasshi : Journal of the Pharmaceutical Society of Japan, 121(6), 379-393 (2001-07-04)
A reaction from various kinds of nitroquinoline with hydroxylamine in potassium hydroxide alkalinity produced a novel product, furazanoquinoline, besides the known amino derivatives. The products obtained were furazano [3,4-f] quinoline (5) from 5-nitroquinoline (1) and 6-nitroquinoline (6), and furazano [3,4-h]
C Chen et al.
Yao xue xue bao = Acta pharmaceutica Sinica, 27(6), 418-422 (1992-01-01)
2-Substituted phenoxy-, 4-methyl-6-methoxy-8-aminoquinolines and 7-methoxy-5-aminoquinoxaline were condensed with 1-phthalimido-bromo-alkane to yield 2-substituted phenoxy-, 4-methyl-6-methoxy-8-(1-phthalimidoalkyl)-aminoquinolines (compounds 7-10 and 15-20) and 7-methoxy-5-(1-phthalimidoalkyl)aminoquinoxalines (28-30) which were subsequently reacted with hydrazine hydrate to give 2-substituted phenoxy-, 4-methyl-6-methoxy-8-(1-aminoalkyl)-aminoquinolines (11-14 and 22-27) and 7-methoxy-5-(1-aminoalkyl) aminoquinoxalines (31-33)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service