132934
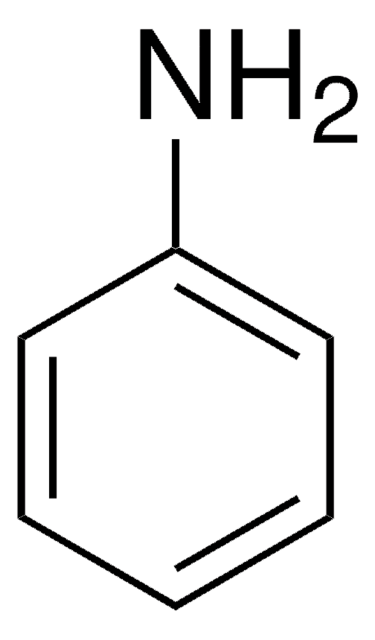
Aniline
ReagentPlus®, 99%
Sinónimos:
Aminobenzene, Benzenamine
About This Item
Productos recomendados
vapor density
3.22 (185 °C, vs air)
Quality Level
vapor pressure
0.7 mmHg ( 25 °C)
product line
ReagentPlus®
assay
99%
form
liquid
autoignition temp.
1139 °F
expl. lim.
11 %
refractive index
n20/D 1.586 (lit.)
pH
8.8 (20 °C, 36 g/L)
bp
184 °C (lit.)
mp
−6 °C (lit.)
solubility
water: soluble
density
1.022 g/mL at 25 °C (lit.)
SMILES string
Nc1ccccc1
InChI
1S/C6H7N/c7-6-4-2-1-3-5-6/h1-5H,7H2
InChI key
PAYRUJLWNCNPSJ-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
General description
Application
Biochem/physiol Actions
Physical properties
Legal Information
signalword
Danger
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 2 - Eye Dam. 1 - Muta. 2 - Skin Sens. 1 - STOT RE 1
target_organs
Blood
Storage Class
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
wgk_germany
WGK 3
flash_point_f
158.0 °F - closed cup
flash_point_c
70 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico