238309
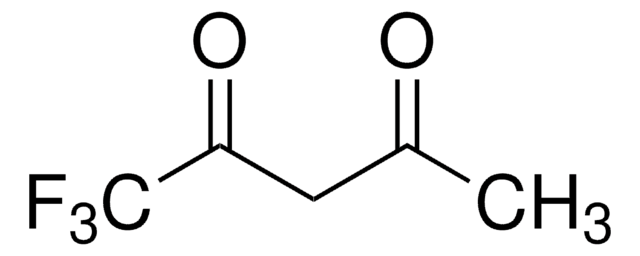
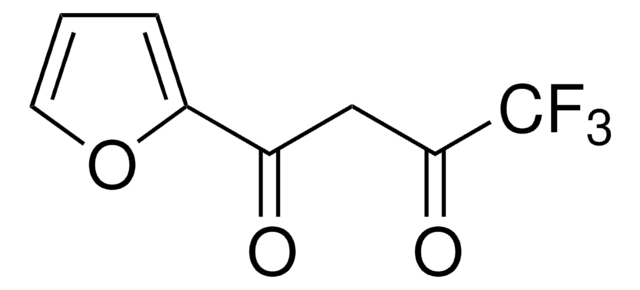
Hexafluoroacetylacetone
98%
Synonym(s):
1,1,1,5,5,5-Hexafluoro-2,4-pentanedione, 1,1,1,5,5,5-Hexafluoroacetylacetone
About This Item
Recommended Products
Quality Level
Assay
98%
form
liquid
refractive index
n20/D 1.332 (lit.)
bp
70-71 °C (lit.)
density
1.47 g/mL at 25 °C (lit.)
functional group
fluoro
ketone
SMILES string
FC(F)(F)C(=O)CC(=O)C(F)(F)F
InChI
1S/C5H2F6O2/c6-4(7,8)2(12)1-3(13)5(9,10)11/h1H2
InChI key
QAMFBRUWYYMMGJ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- Antipsychotic medications and sleep problems in patients with schizophrenia: This research investigates the effects of antipsychotics, including aripiprazole, on sleep disturbances in schizophrenia patients, highlighting its pharmacodynamic actions and contributing to a deeper understanding of aripiprazole′s clinical applications in mental health disorders (Cederlöf et al., 2024).
accessory
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Eye Dam. 1 - Flam. Liq. 3 - Skin Corr. 1B
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
89.6 °F - closed cup
Flash Point(C)
32 °C - closed cup
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 238309-5G | 4061838786838 |
| 238309-5KG | |
| 238309-25G | 4061838786821 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service