추천 제품
Grade
reagent grade
제품 라인
Vetec™
분석
99%
양식
powder
mp
88-92 °C (lit.)
저장 온도
2-8°C
SMILES string
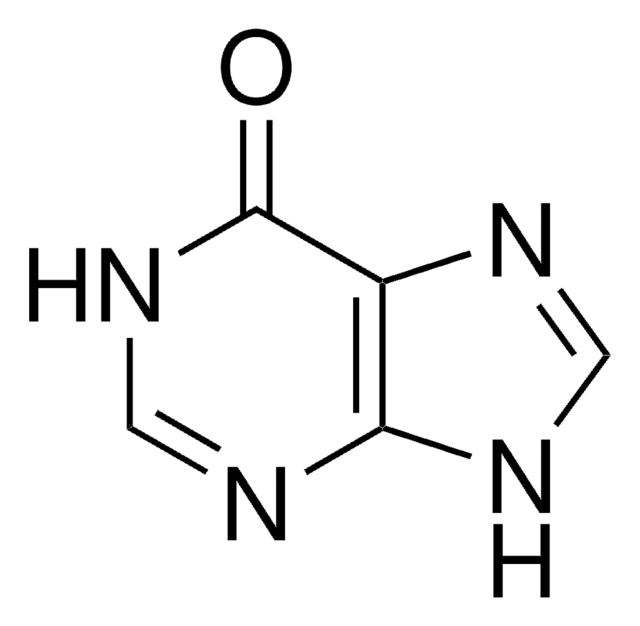
OC[C@@H](O)[C@@H](O)[C@@H](O)C([H])=O
InChI
1S/C5H10O5/c6-1-3(8)5(10)4(9)2-7/h1,3-5,7-10H,2H2/t3-,4+,5-/m0/s1
InChI key
PYMYPHUHKUWMLA-LMVFSUKVSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
애플리케이션
- Conditions and Mechanism of Formation of the Maillard Reaction Pigment, Furpenthiazinate, in a Model System and in Some Acid Hydrolyzates of Foods and its Biological Properties.: This article investigates the conditions and mechanisms under which ᴅ-(−)-Ribose participates in the Maillard reaction to form bioactive pigments like Furpenthiazinate. It details the implications for food chemistry and the nutritional properties of processed foods (Noda et al., 2024).
기타 정보
To gain a comprehensive understanding of our extensive range of Monosaccharides for your research, we encourage you to visit our Carbohydrates Category page.
법적 정보
Vetec is a trademark of Merck KGaA, Darmstadt, Germany
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
이미 열람한 고객
Thomas L Willett et al.
Bone, 52(2), 611-622 (2012-11-28)
Non-enzymatic glycation (NEG) and advanced glycation endproducts (AGEs) may contribute to bone fragility in various diseases, ageing, and other conditions by modifying bone collagen and causing degraded mechanical properties. In this study, we sought to further understand how collagen modification
Lusani Norah Vhangani et al.
Food chemistry, 137(1-4), 92-98 (2012-12-04)
Maillard reaction products (MRPs) were prepared from aqueous ribose-lysine (RL) and fructose-lysine (FL) model systems at pH 9, heated at 60, 80 and 120 °C for 15, 60 and 120 min. Browning intensity (BI) and pH reduction were monitored throughout
Raman K Sharma et al.
Bioorganic & medicinal chemistry, 20(23), 6821-6830 (2012-10-27)
A series of peracetylated O-aryl α,β-d-ribofuranosides have been synthesized and an efficient biocatalytic methodology has been developed for the separation of their anomers which was otherwise almost impossible by column chromatographic or other techniques. The incubation of 2,3,5-tri-O-acetyl-1-O-aryl-α,β-d-ribofuranoside with Lipozyme®
Carine Baraguey et al.
Organic & biomolecular chemistry, 11(16), 2638-2647 (2013-03-05)
The pivaloyloxymethyl (PivOM) group is a biolabile 2'-O-ribose protection that is under development in a prodrug-based approach for siRNA applications. Besides an expected cellular uptake, nucleic acid sequences carrying PivOM showed also increased nuclease resistance and, in most cases, an
Anders Virtanen et al.
Critical reviews in biochemistry and molecular biology, 48(2), 192-209 (2013-03-19)
Deadenylation of eukaryotic mRNA is a mechanism critical for mRNA function by influencing mRNA turnover and efficiency of protein synthesis. Here, we review poly(A)-specific ribonuclease (PARN), which is one of the biochemically best characterized deadenylases. PARN is unique among the
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.