1435003
USP
Methylprednisolone
United States Pharmacopeia (USP) Reference Standard
동의어(들):
6α-Methylprednisolone, 11β,17α,21-Trihydroxy-6α-methyl-1,4-pregnadiene-3,20-dione, 6α-Methyl-11β,17α,21-trihydroxy-1,4-pregnadiene-3,20-dione, Medrol, Medrone
About This Item
추천 제품
Grade
pharmaceutical primary standard
API family
methylprednisolone
제조업체/상표
USP
응용 분야
pharmaceutical (small molecule)
형식
neat
SMILES string
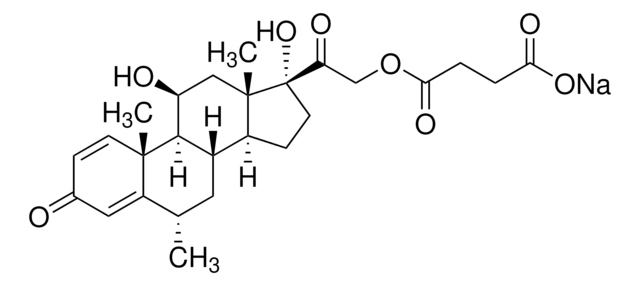
[H][C@@]12C[C@H](C)C3=CC(=O)C=C[C@]3(C)[C@@]1([H])[C@@H](O)C[C@@]4(C)[C@@]2([H])CC[C@]4(O)C(=O)CO
InChI
1S/C22H30O5/c1-12-8-14-15-5-7-22(27,18(26)11-23)21(15,3)10-17(25)19(14)20(2)6-4-13(24)9-16(12)20/h4,6,9,12,14-15,17,19,23,25,27H,5,7-8,10-11H2,1-3H3/t12-,14-,15-,17-,19+,20-,21-,22-/m0/s1
InChI key
VHRSUDSXCMQTMA-PJHHCJLFSA-N
유전자 정보
human ... NR3C1(2908)
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
애플리케이션
- Methylprednisolone Sodium Succinate for Injection
- Methylprednisolone Tablets
분석 메모
기타 정보
관련 제품
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Repr. 1B - STOT RE 2
표적 기관
Adrenal gland,Immune system
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 2
가장 최신 버전 중 하나를 선택하세요:
이미 열람한 고객
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.