T4580
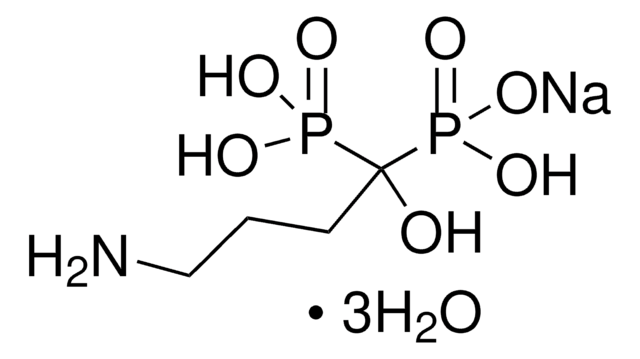
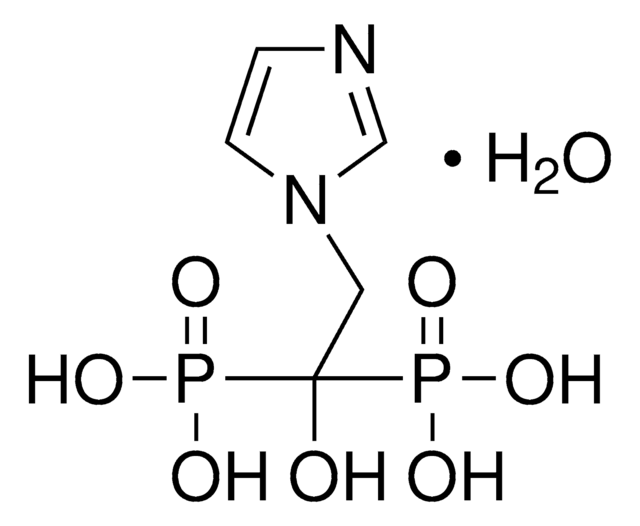
Tiludronate disodium salt hydrate
≥98% (HPLC)
동의어(들):
Skelid, Tiludronic acid disodium, [[(4-Chlorophenyl)thio]methylene]bisphosphonic acid disodium salt hydrate
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C7H7ClNa2O6P2S · xH2O
CAS Number:
Molecular Weight:
362.57 (anhydrous basis)
MDL number:
UNSPSC 코드:
12352200
PubChem Substance ID:
NACRES:
NA.77
추천 제품
분석
≥98% (HPLC)
양식
powder
저장 조건
desiccated
색상
white to off-white
solubility
H2O: >10 mg/mL, clear
주관자
Sanofi Aventis
SMILES string
O.[Na+].[Na+].OP([O-])(=O)C(Sc1ccc(Cl)cc1)P(O)([O-])=O
InChI
1S/C7H9ClO6P2S.2Na.H2O/c8-5-1-3-6(4-2-5)17-7(15(9,10)11)16(12,13)14;;;/h1-4,7H,(H2,9,10,11)(H2,12,13,14);;;1H2/q;2*+1;/p-2
InChI key
SZVJRJRMQCKFON-UHFFFAOYSA-L
생화학적/생리학적 작용
Tiludronate is a bisphosphonate bone resorption inhibitor. Tiludronate inhibits protein-tyrosine-phosphatase, leading to detachment of osteoclasts from the bone surface. It also inhibits the osteoclastic proton pump and is used to treat Paget′s disease.
특징 및 장점
This compound was developed by Sanofi Aventis. To browse the list of other pharma-developed compounds and Approved Drugs/Drug Candidates, click here.
제조 메모
Tiludronate disodium salt hydrate is soluble in DMSO at a concentration that is greater than or equal to 10 mg/ml.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
이미 열람한 고객
L Alvarez et al.
Rheumatology (Oxford, England), 43(7), 869-874 (2004-04-01)
To monitor the long-term evolution of Paget's disease activity after treatment with tiludronate by using serum total alkaline phosphatase (TAP) and more sensitive markers such as bone alkaline phosphatase (BAP), procollagen type I N propeptide (PINP) and urinary N-terminal cross-linking
Virginie Coudry et al.
American journal of veterinary research, 68(3), 329-337 (2007-03-03)
To evaluate the efficacy of tiludronate for the treatment of horses with signs of pain associated with lesions of the thoracolumbar vertebral column. 29 horses with clinical manifestations of pain associated with lesions of the thoracolumbar vertebral column and abnormal
C Delguste et al.
Journal of veterinary pharmacology and therapeutics, 31(2), 108-116 (2008-03-01)
Bioavailability and pharmacological effects of tiludronate were compared when administered as an intravenous (i.v.) bolus at a dosage of 0.1 mg/kg body weight (b.w.) once daily for 10 consecutive days (group 1, n = 6) and as a single constant
C Delguste et al.
Bone, 41(3), 414-421 (2007-07-03)
Tiludronate, a bisphosphonate, has recently been introduced in veterinary medicine to treat orthopedic conditions in the horse. This study was designed to evaluate its effects on biochemical biomarkers of bone metabolism and on bone density and structure in an experimental
Katja F Duesterdieck-Zellmer et al.
American journal of veterinary research, 73(10), 1530-1539 (2012-09-28)
To determine concentration-dependent effects of tiludronate on cartilage explants incubated with or without recombinant equine interleukin-1β (rEq IL-1). Articular cartilage explants from the femorotibial joints of 3 young adult horses. Cartilage explants were incubated with 1 of 6 concentrations (0
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.