SML2417
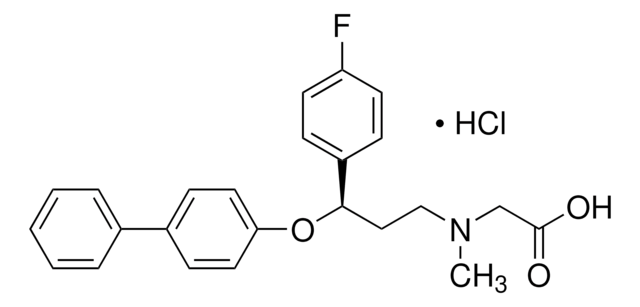
ORG 25543 Hydrochloride
≥95% (HPLC)
동의어(들):
4-Benzyloxy-3,5-dimethoxy-N-[(1-dimethylaminocyclopentyl)methyl]benzamide hydrochloride, N-[[1-(Dimethylamino)cyclopentyl]methyl]-3,5-dimethoxy-4-(phenylmethoxy)benzamide hydrochloride, ORG 25,543 hydrochloride, ORG 25543 hydrochloride, ORG-25543 hydrochloride
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
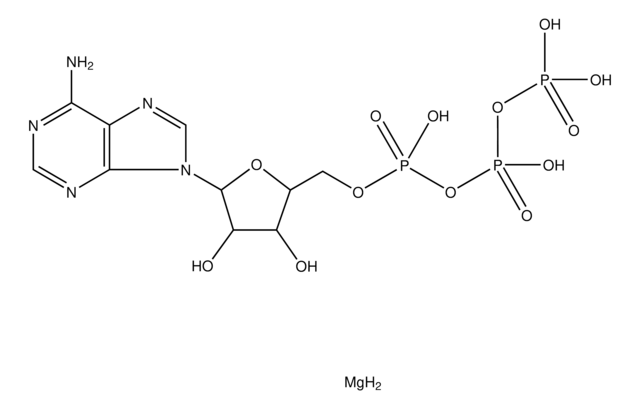
C24H32N2O4·HCl
CAS Number:
Molecular Weight:
448.98
MDL number:
UNSPSC 코드:
12352200
NACRES:
NA.77
추천 제품
분석
≥95% (HPLC)
양식
powder
저장 조건
desiccated
색상
white to beige
solubility
DMSO: 2 mg/mL, clear
저장 온도
room temp
SMILES string
Cl.N(C3(CCCC3)CNC(=O)c1cc(c(c(c1)OC)OCc2ccccc2)OC)(C)C
InChI
1S/C24H32N2O4.ClH/c1-26(2)24(12-8-9-13-24)17-25-23(27)19-14-20(28-3)22(21(15-19)29-4)30-16-18-10-6-5-7-11-18;/h5-7,10-11,14-15H,8-9,12-13,16-17H2,1-4H3,(H,25,27);1H
InChI key
NIPQJILJYQVZJR-UHFFFAOYSA-N
생화학적/생리학적 작용
Brain-penetrant, high-affinity, potent and selective glycine transporter 2 (GlyT-2; GlyT2) inhibitor with higher potency and selectivity than ALX-1393.
ORG 25543 is a brain-penetrant (free brain/plasma ratio = 0.53; 35 min post 2 or 20 mg/kg i.v. in mice), high-affinity, potent and selective glycine transporter 2 (GlyT-2; GlyT2) inhibitor (human & mouse pIC50 = 7.9/ GlyT2 vs <4/GlyT1) with great selectivity over a panel of 56 receptor and channel proteins. ORG 25543 is more potent and selective than the brain-impermeable ALX-1393 (GlyT2/GlyT1 pIC50 = 7.1/5.4) and exhibits high in vivo efficacy in a murine diabetic neuropathic pain model (ED50 = 0.07-0.16 mg/kg i.v.; Emin = 0.01 mg/kg). ORG 25543 is practically irreversible due to its tight-binding nature, suboptimal dosage should be applied in vivo to allow low target occupancy only and minimize acute toxicity.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
A Mingorance-Le Meur et al.
British journal of pharmacology, 170(5), 1053-1063 (2013-08-22)
Available medications for chronic pain provide only partial relief and often cause unacceptable side effects. There is therefore a need for novel molecular targets to develop new therapeutics with improved efficacy and tolerability. Despite encouraging efficacy data in rodents with
Katsuya Morita et al.
The Journal of pharmacology and experimental therapeutics, 326(2), 633-645 (2008-05-02)
Neuropathic pain is refractory against conventional analgesics, and thus novel medicaments are desired for the treatment. Glycinergic neurons are localized in specific brain regions, including the spinal cord, where they play an important role in the regulation of pain signal
M A Gradwell et al.
The Journal of physiology, 595(23), 7185-7202 (2017-09-15)
Spinal parvalbumin-expressing interneurons have been identified as a critical source of inhibition to regulate sensory thresholds by gating mechanical inputs in the dorsal horn. This study assessed the inhibitory regulation of the parvalbumin-expressing interneurons, showing that synaptic and tonic glycinergic
Cristina Romei et al.
Brain research bulletin, 127, 100-110 (2016-10-28)
There is increasing evidence for the neuronal coexistence of classical transmitters. Implications in favor of cotransmission have often been represented by the identification, in the same neuron, of the putative cotransmitters, their synthetic enzymes and/or their vesicular transporters. In contrast
Cristina Romei et al.
Journal of neurochemistry, 119(1), 50-63 (2011-07-28)
Glycine release provoked by ion dysregulations typical of some neuropathological conditions was analyzed in cerebellar synaptosomes selectively pre-labelled with [³H]glycine through GlyT2 transporters and exposed in superfusion to KCl, 4-aminopyridine (4-AP) or veratridine. The overflows caused by relatively low concentrations
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.

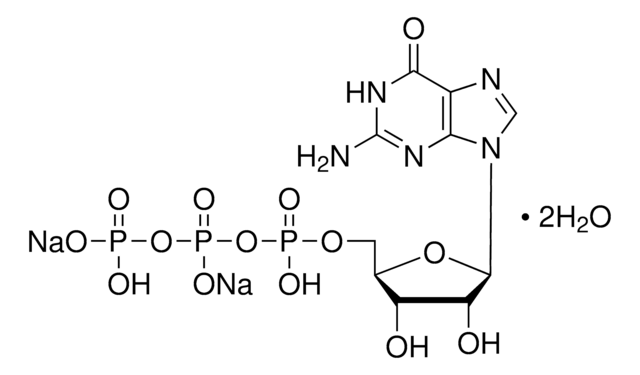

![Guanosine 5′-[β,γ-imido]triphosphate trisodium salt hydrate ≥85% (HPLC), powder](/deepweb/assets/sigmaaldrich/product/structures/204/494/05808804-1ca7-44bf-b6c5-d4934dc7cb85/640/05808804-1ca7-44bf-b6c5-d4934dc7cb85.png)