SML0477
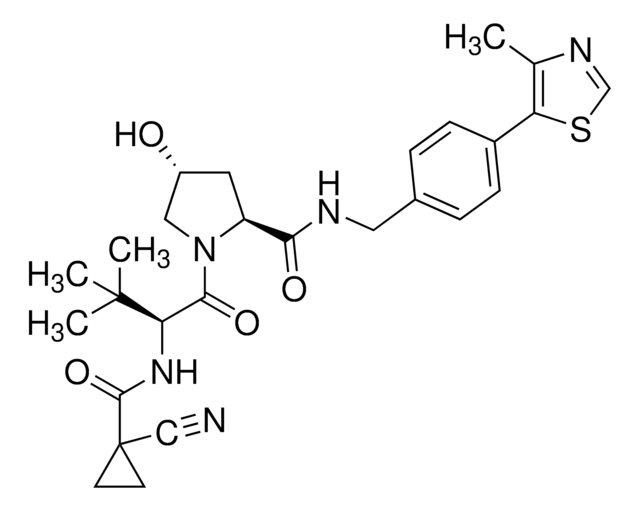
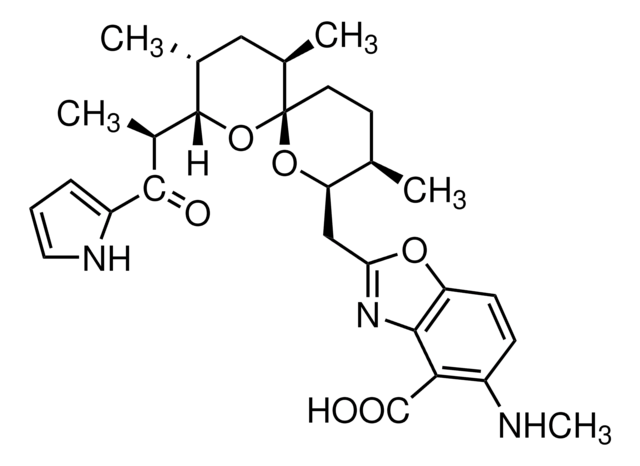
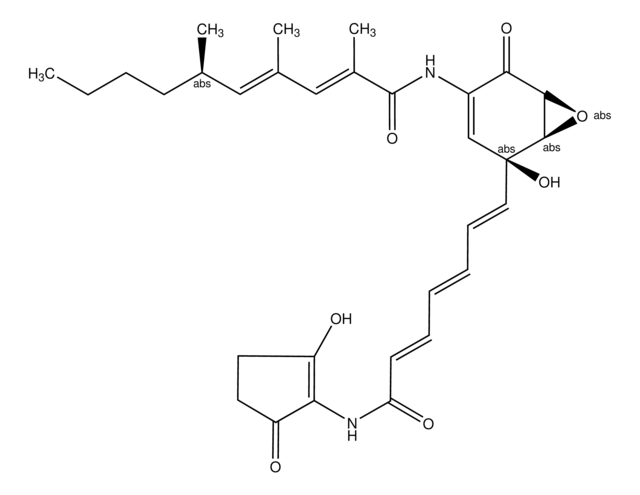
Echinomycin
≥98% (HPLC)
동의어(들):
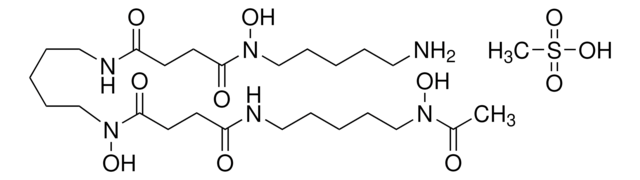
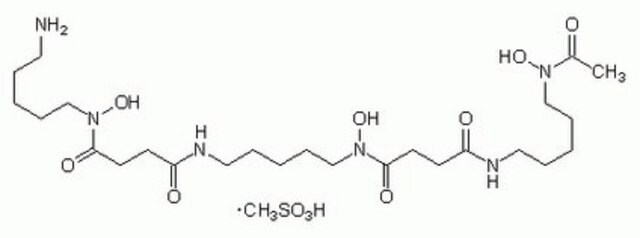
N-(2-Quinoxalinylcarbonyl)-O-[N-(2-quinoxalinylcarbonyl)-D-seryl-L-alanyl-3-mercapto-N,S-dimethylcysteinyl-N-methyl-L-valyl]-D-seryl-L-alanyl-N-methylcysteinyl-N-methyl L-valine (81)-lactone cyclic (37)-thioester, Quinomycin A
About This Item
추천 제품
Quality Level
분석
≥98% (HPLC)
양식
powder
저장 조건
desiccated
색상
white to beige
배송 상태
dry ice
저장 온도
−20°C
InChI
1S/C51H64N12O12S2/c1-25(2)38-49(72)74-22-36(59-42(65)34-21-53-30-17-13-15-19-32(30)57-34)44(67)55-28(6)46(69)63(10)40-48(71)62(9)39(26(3)4)50(73)75-23-35(58-41(64)33-20-52-29-16-12-14-18-31(29)56-33)43(66)54-27(5)45(68)60(7)37(47(70)61(38)8)24-77-51(40)76-11/h12-21,25-28,35-40,51H,22-24H2,1-11H3,(H,54,66)(H,55,67)(H,58,64)(H,59,65)
InChI key
AUJXLBOHYWTPFV-UHFFFAOYSA-N
애플리케이션
생화학적/생리학적 작용
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Repr. 2
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
이미 열람한 고객
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.