S3626
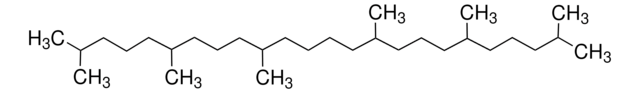
Squalene
≥98%, liquid
동의어(들):
2,6,10,15,19,23-Hexamethyl-2,6,10,14,18,22-tetracosahexaene
About This Item
추천 제품
Quality Level
분석
≥98%
형태
liquid
색상
light yellow
refractive index
n20/D 1.494 (lit.)
bp
285 °C/25 mmHg (lit.)
mp
−75 °C (lit.)
density
0.858 g/mL at 25 °C (lit.)
응용 분야
metabolomics
vitamins, nutraceuticals, and natural products
저장 온도
2-8°C
SMILES string
CC(C)=CCCC(C)=CCCC(C)=CCC\C=C(/C)CCC=C(C)CCC=C(C)C
InChI
1S/C30H50/c1-25(2)15-11-19-29(7)23-13-21-27(5)17-9-10-18-28(6)22-14-24-30(8)20-12-16-26(3)4/h15-18,23-24H,9-14,19-22H2,1-8H3/b27-17+,28-18+,29-23+,30-24+
InChI key
YYGNTYWPHWGJRM-AAJYLUCBSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
애플리케이션
- as a standard for lipid identification and quantification
- in the isolation of macrophages for parasite incubation
- as a standard for the quantification of squalene in squalene analysis of oil samples
생화학적/생리학적 작용
신호어
Danger
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Asp. Tox. 1
Storage Class Code
10 - Combustible liquids
WGK
WGK 2
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
문서
Terpenes comprise the largest and most diverse class of secondary metabolites; approximately 55,000 compounds have been identified to date.
Biosynthesis of cholesterol generally takes place in the endoplasmic reticulum of hepatic cells and begins with acetyl- CoA, which is mainly derived from an oxidation reaction in the mitochondria. Acetyl-CoA and acetoacetyl-CoA are converted to 3-hydroxy- 3-methylglutaryl-CoA (HMG-CoA) by HMG-CoA synthase.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.