크기 선택
모든 사진(1)
크기 선택
보기 변경
About This Item
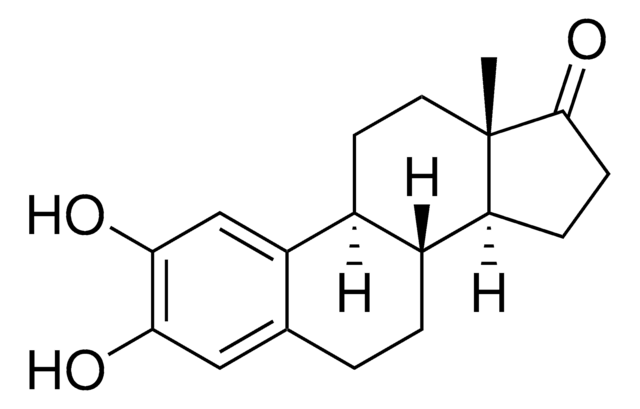
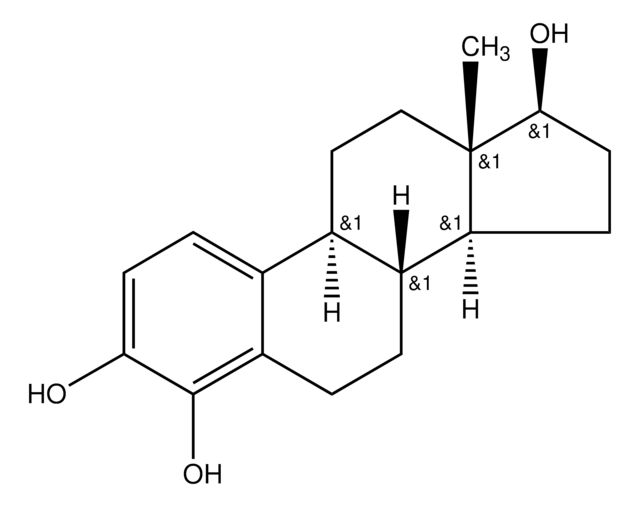
실험식(Hill 표기법):
C18H22O3
CAS Number:
Molecular Weight:
286.37
MDL number:
UNSPSC 코드:
51111800
PubChem Substance ID:
NACRES:
NA.77
추천 제품
무균
non-sterile
Quality Level
분석
≥90% (HPLC)
양식
powder
solubility
methanol: 10 mg/mL, clear, colorless to faintly brownish-yellow
배송 상태
ambient
저장 온도
−20°C
SMILES string
[H][C@]12CC[C@]3(C)C(=O)CC[C@@]3([H])[C@]1([H])CCc4c(O)c(O)ccc24
InChI
1S/C18H22O3/c1-18-9-8-11-10-4-6-15(19)17(21)13(10)3-2-12(11)14(18)5-7-16(18)20/h4,6,11-12,14,19,21H,2-3,5,7-9H2,1H3/t11-,12-,14+,18+/m1/s1
InChI key
XQZVQQZZOVBNLU-QDTBLXIISA-N
생화학적/생리학적 작용
4-Hydroxyestrone is an endogenous estrogen metabolite, which exhibits a strong neuroprotective effect against oxidative damage. It also provides effective protection against kanic acid-induced hippocampal oxidative damage in rats when compared to 17β-estradiol. 4-Hydroxyestrone regulates the angiogenic process during corpus luteum formation. It might be involved in an increased risk of cancer. 4-Hydroxyestrone is found in the early and mid-luteal phases.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
199.9 °F - closed cup
Flash Point (°C)
93.3 °C - closed cup
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
Shelley A Gestl et al.
The American journal of pathology, 160(4), 1467-1479 (2002-04-12)
Glucuronidation, mediated by UDP-glucuronosyltransferases (UGTs), affects the actions and disposition of diverse endo- and xenobiotics. In the case of catecholestrogens (CEs), glucuronidation is likely to block their oxidation to quinone estrogens that are the putative mediators of CEs' actions as
Y Chen et al.
Chemical research in toxicology, 13(5), 342-350 (2000-05-17)
Estrogen replacement therapy has been correlated with an increased risk of developing breast or endometrial cancer. 4-Hydroxyequilenin (4-OHEN) is a catechol metabolite of equilenin which is a minor component of the estrogen replacement formulation marketed under the name of Premarin
B T Zhu et al.
Endocrinology, 135(5), 1772-1779 (1994-11-01)
As part of an ongoing investigation of the role of metabolic activation of estrogens in the genesis of cancers such as estrogen-induced renal tumors in hamsters, we have 1) determined steroid-17 beta-oxidoreductase activity of microsomes and cytosol prepared from hamster
X Xu et al.
The Journal of clinical endocrinology and metabolism, 84(11), 3914-3918 (1999-11-24)
Endogenous estrogen metabolism may play an important role in the pathogenesis of hormone-related cancers, most notably breast cancer. Despite the importance of estrogen metabolism, little is known about estrogen metabolite profiles during different phases of the menstrual cycle. This study
E Pisha et al.
Chemical research in toxicology, 14(1), 82-90 (2001-02-15)
Estrogen replacement therapy has been correlated with an increased risk of developing hormone-dependent cancers. 4-Hydroxyequilenin (4-OHEN) is a catechol metabolite of equilenin and equilin which are components of the estrogen replacement formulation marketed under the name of Premarin (Wyeth-Ayerst). Previously
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.