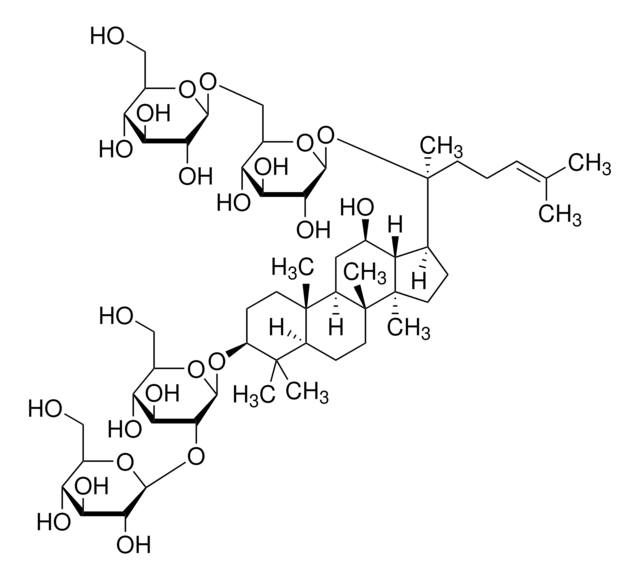
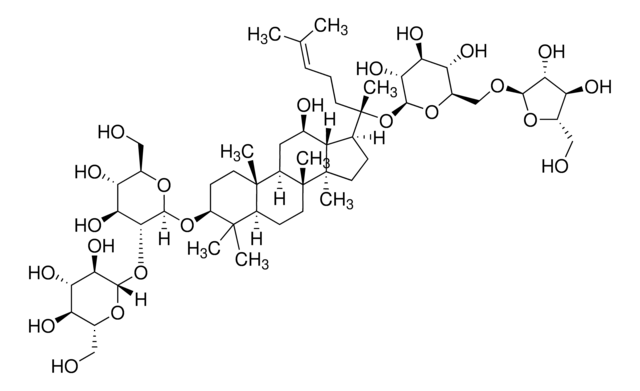
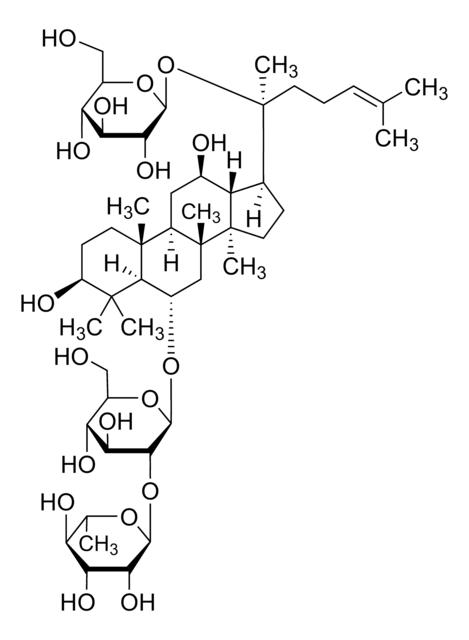
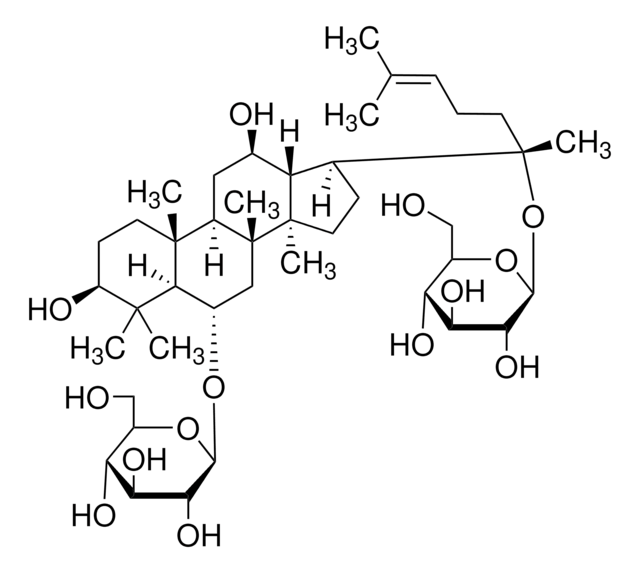
추천 제품
생물학적 소스
plant root (Panax ginseng)
Quality Level
분석
≥98% (HPLC)
양식
powder
기술
HPLC: suitable
저장 온도
2-8°C
SMILES string
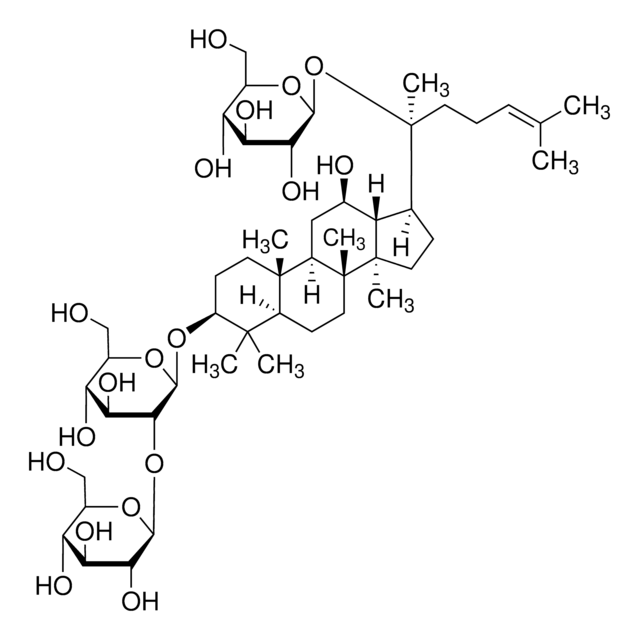
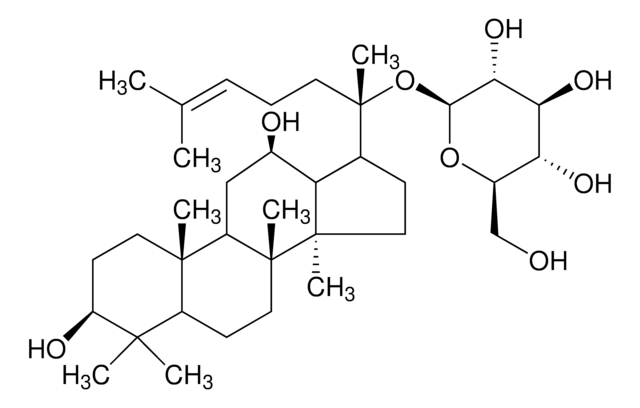
C\C(C)=C\CCC(C)(OC1OC(COC2OC(CO)C(O)C2O)C(O)C(O)C1O)C3CCC4(C)C3C(O)CC5C6(C)CCC(OC7OC(CO)C(O)C(O)C7OC8OC(CO)C(O)C(O)C8O)C(C)(C)C6CCC45C
InChI
1S/C53H90O22/c1-23(2)10-9-14-53(8,75-47-43(67)39(63)37(61)29(72-47)22-68-45-41(65)36(60)28(21-56)69-45)24-11-16-52(7)33(24)25(57)18-31-50(5)15-13-32(49(3,4)30(50)12-17-51(31,52)6)73-48-44(40(64)35(59)27(20-55)71-48)74-46-42(66)38(62)34(58)26(19-54)70-46/h10,24-48,54-67H,9,11-22H2,1-8H3
InChI key
JDCPEKQWFDWQLI-UHFFFAOYSA-N
생화학적/생리학적 작용
Ginsenoside-Rc exhibits anti-proliferative effects on human breast cancer cell lines and can induce expreβion of the leucine-zipper transcription factor c-fos.
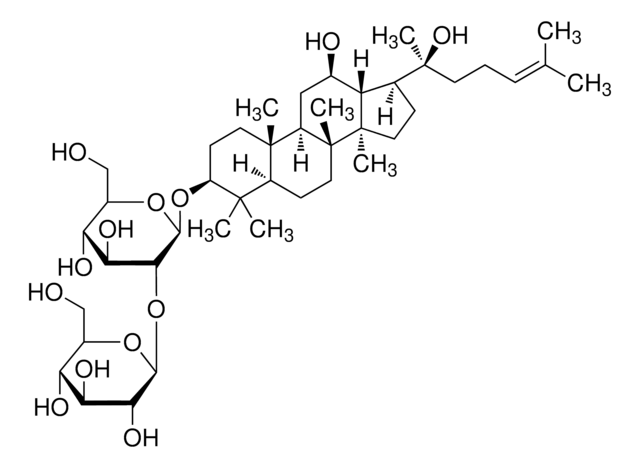
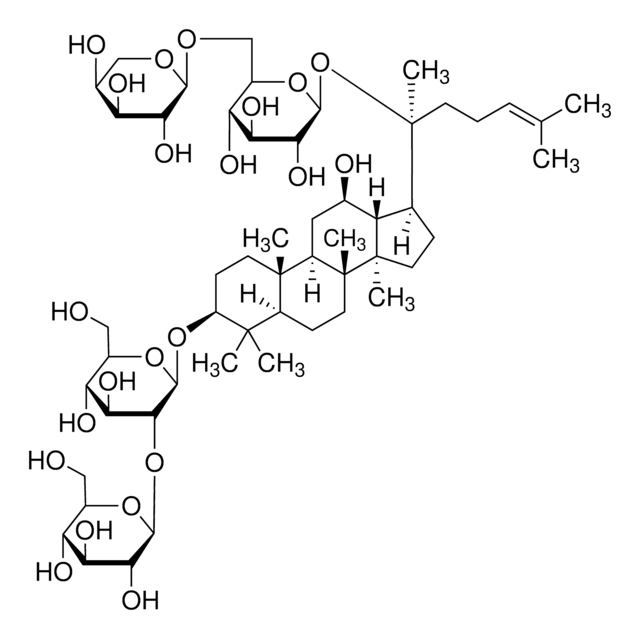
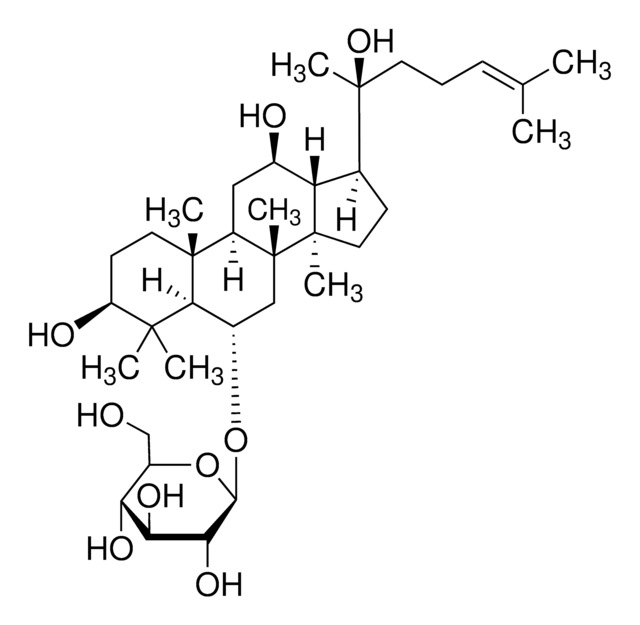
Triterpene saponin found in ginseng.
기타 정보
To gain a comprehensive understanding of our extensive range of Polysaccharides for your research, we encourage you to visit our Carbohydrates Category page.
신호어
Warning
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
Louis Y Chan et al.
Reproductive toxicology (Elmsford, N.Y.), 19(1), 131-134 (2004-09-01)
Pregnant women commonly consume ginseng. However, there is little data concerning the effects of ginseng on early pregnancy. Rat embryos were exposed in vitro to different concentrations of Rc and Re from day 9.5 to day 11.5 after conception. Embryos
Myoung-Su Lee et al.
Journal of ethnopharmacology, 127(3), 771-776 (2009-12-08)
Panax ginseng and its major component, ginsenosides, are widely used for the prevention of various disorders in oriental medicine. To evaluate the effect of ginsenoside Rc (Rc), one of the active constituents in Panax ginseng, on glucose uptake in C2C12
Ron Luchtefeld et al.
Journal of agricultural and food chemistry, 52(16), 4953-4956 (2004-08-05)
A method based on high-performance liquid chromatography (HPLC) and negative ion electrospray mass spectrometry (LC-MS) has been used to determine ginsenosides Rb1, Rc, and Re in six different samples of ginseng. These included a liquid extract, capsules, tea bags, and
T B Ng et al.
Journal of ethnopharmacology, 16(2-3), 191-199 (1986-06-01)
Ginsenosides Rb2, Rc and Rg1 suppressed corticotropin-induced, dibutyryl cyclic AMP-induced and epinephrine-induced lipolysis with the relative potencies Rb2 greater than Rc greater than Rg1. The inhibition of corticotropin-induced lipolysis by ginsenoside Rg1 could not be overcome by increasing the dose
Young Joo Lee et al.
Archives of pharmacal research, 26(1), 53-57 (2003-02-06)
We have found that ginsenoside Rc and Re induce c-fos in MCF-7 human breast carcinoma cells at both the mRNA and protein levels. However, neither ginsenoside activated the expression of reporter gene under the control of AP-1/TPA response elements. We
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.