추천 제품
제품명
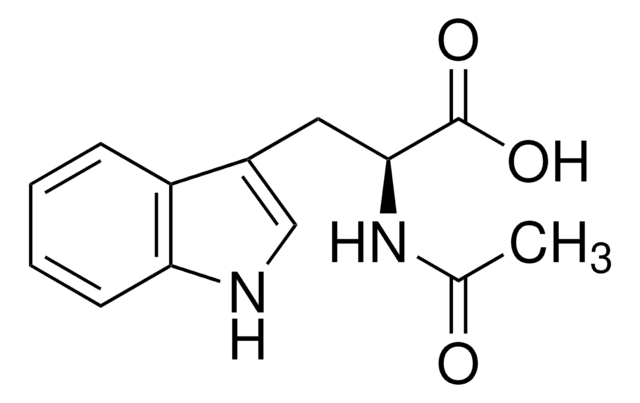
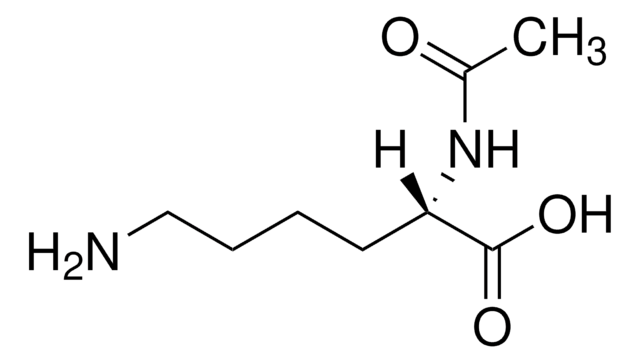
N-Acetyl-L-tryptophan,
분석
≥99% (TLC)
Quality Level
양식
powder
기술
ligand binding assay: suitable
색상
white to off-white
저장 온도
2-8°C
SMILES string
CC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(O)=O
InChI
1S/C13H14N2O3/c1-8(16)15-12(13(17)18)6-9-7-14-11-5-3-2-4-10(9)11/h2-5,7,12,14H,6H2,1H3,(H,15,16)(H,17,18)/t12-/m0/s1
InChI key
DZTHIGRZJZPRDV-LBPRGKRZSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
생화학적/생리학적 작용
N-Acetyl-L-tryptophan (NAT, Ac-Trp-OH) is used a substance P NK1 tachykinin receptor antagonist. N-Acetyl-L-tryptophan is also used as a competitive inhibitor to identify, differentiate and characterized tryptophanase(s).
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
이미 열람한 고객
James J Donkin et al.
Journal of neurotrauma, 28(2), 217-224 (2010-12-24)
Previous studies have demonstrated that the compound N-acetyl-L-tryptophan (NAT) reduces brain edema and improves functional outcome following traumatic brain injury (TBI). In this study we examined whether this effect was mediated via the neurokinin-1 receptor, and whether there was an
New tryptophanase inhibitors: towards prevention of bacterial biofilm formation.
Scherzer R, Gdalevsky GY, et al.
Journal of Enzyme Inhibition, 24, 350-355 (2009)
Kate M Lewis et al.
Anti-cancer drugs, 24(4), 344-354 (2013-02-15)
Emend, an NK1 antagonist, and dexamethasone are used to treat complications associated with metastatic brain tumours and their treatment. It has been suggested that these agents exert anticancer effects apart from their current use. The effects of the NK1 antagonists
Toshinori Suzuki et al.
Free radical biology & medicine, 37(5), 671-681 (2004-08-04)
Proteins are targets of reactive nitrogen species such as peroxynitrite and nitrogen dioxide. Among the various amino acids in proteins, tryptophan residues are especially susceptible to attack by reactive nitrogen species. We carried out experiments on the reactions of peroxynitrite
Renée J Turner et al.
Brain research, 1393, 84-90 (2011-04-07)
Previous studies have suggested that substance P (SP) plays a critical role in the development of brain oedema and functional deficits following traumatic brain injury and that SP receptor antagonism may improve outcome. No studies have described such a role
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.