모든 사진(1)
About This Item
실험식(Hill 표기법):
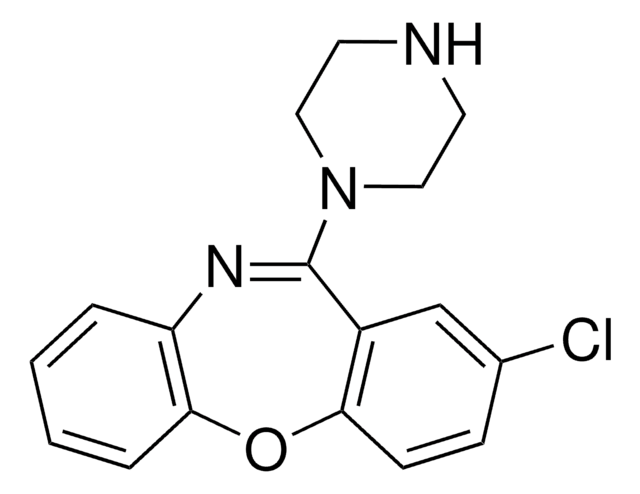
C17H16ClN3O
CAS Number:
Molecular Weight:
313.78
EC Number:
MDL number:
UNSPSC 코드:
12352200
PubChem Substance ID:
NACRES:
NA.77
추천 제품
형태
powder
Quality Level
solubility
methanol: soluble
주관자
Wyeth
SMILES string
Clc1ccc2Oc3ccccc3N=C(N4CCNCC4)c2c1
InChI
1S/C17H16ClN3O/c18-12-5-6-15-13(11-12)17(21-9-7-19-8-10-21)20-14-3-1-2-4-16(14)22-15/h1-6,11,19H,7-10H2
InChI key
QWGDMFLQWFTERH-UHFFFAOYSA-N
유전자 정보
human ... DRD1(1812) , DRD2(1813) , DRD3(1814) , DRD4(1815) , DRD5(1816) , HTR2A(3356) , HTR2B(3357) , HTR2C(3358) , SLC6A2(6530) , SLC6A4(6532)
유사한 제품을 찾으십니까? 방문 제품 비교 안내
애플리케이션
Amoxapine has been used as an antidepressant drug to test its effect on locomotion and egg release in response to gain-of-functional mutations in potassium (K+) channels (unc-58) of C. elegans. It has also been used as an antipsychotic drug to test its effect on the viability of glioblastoma cells.
생화학적/생리학적 작용
Amoxapine, a structural analog of clozapine, is a human ether a-go-go (hERG) channel blocker. It is also an N-methylated metabolite of loxapine. It is a tricyclic antidepressant that inhibits the uptake of norepinephrine and blocks 5- hydroxytryptamine (HT2 ) serotonergic receptors.
특징 및 장점
This compound is featured on the Biogenic Amine Transporters page of the Handbook of Receptor Classification and Signal Transduction. To browse other handbook pages, click here.
This compound was developed by Wyeth. To browse the list of other pharma-developed compounds and Approved Drugs/Drug Candidates, click here.
신호어
Warning
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
S G Jue et al.
Drugs, 24(1), 1-23 (1982-07-01)
Amoxapine is an N-demethylated dibenzoxazepine closely related in the neuroleptic loxapine. Its tricyclic structure appears to give it antidepressant properties resembling imipramine and amitriptyline. In uncontrolled trials it was shown to have antidepressant activity in usual doses up to 200
S Kapur et al.
Biological psychiatry, 45(9), 1217-1220 (1999-05-20)
All currently available atypical antipsychotics have, at clinically relevant doses: i) high serotonin (5-HT)2 occupancy; ii) greater 5-HT2 than dopamine (D)2 occupancy; and iii) a higher incidence of extrapyramidal side effects when their D2 occupancy exceeds 80%. A review of
Imran B Chaudhry et al.
Journal of clinical psychopharmacology, 27(6), 575-581 (2007-11-16)
It has been proposed that the lack of extrapyramidal side effects of atypical antipsychotic drugs is caused by their fast dissociation or low affinity for the D2 receptor or their concomitant high affinity for other receptors, for example, 5HT2 and
Yan-Lin He et al.
The Journal of pharmacology and experimental therapeutics, 332(2), 437-445 (2009-11-17)
Ion channels are known to be modulated by antidepressant drugs, but the molecular mechanisms are not known. We have shown that the antidepressant drug amoxapine suppresses rectifier outward K(+) (I(K)) currents in mouse cortical neurons. At a concentration of 10
F Jenck et al.
Progress in neuro-psychopharmacology & biological psychiatry, 18(3), 563-574 (1994-05-01)
A variety of antidepressants of different chemical classes were tested for their in vivo and in vitro activity at 5-HT1C receptors in the brain. Conventional tricyclic antidepressants (imipramine, desipramine, maprotiline, clomipramine, trimipramine, amitriptyline, nortriptyline, doxepin, amoxapine, oxaprotiline) and two atypical
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.