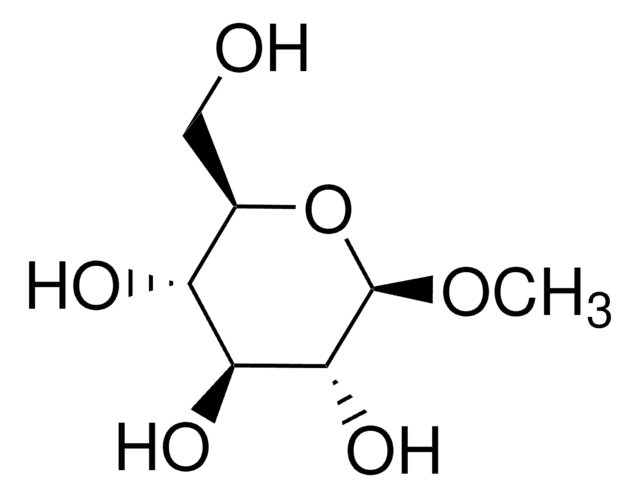
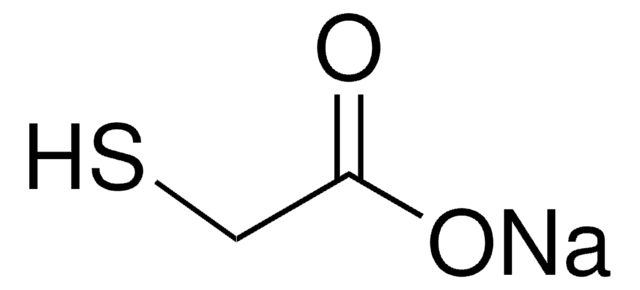
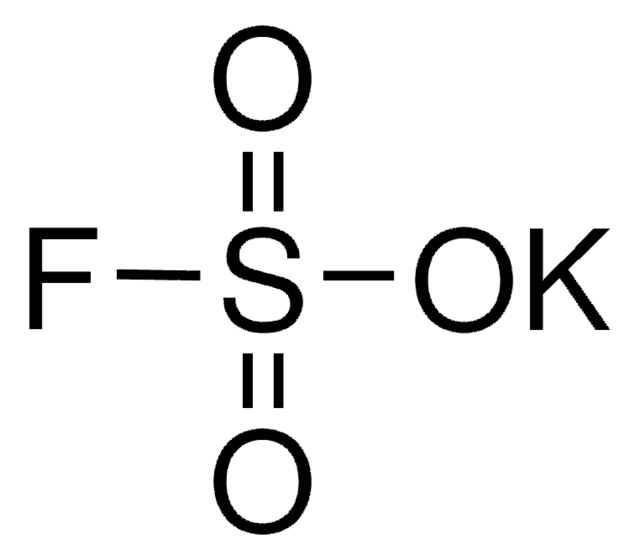
추천 제품
일반 설명
Potassium sulfate is an inorganic salt that can be prepared by reacting phosphogypsum and potassium chloride. It forms needlelike mullite particles on heating with aluminum sulfate (Al2(SO4)3) and silicon dioxide (SiO2). Its application to the soil has been reported to minimize the bronzing of rice plants. Its surface integration growth kinetics has been obtained in the temperature range of 20-50°C. The theoretical heat capacity curve of potassium sulfate in vapor phase has been obtained.
애플리케이션
Potassium sulfate may be used as a sulfonating agent in the preparation of 4′-dibenzo-18-crown-6-sulfonic acid via sulfonation of dibenzo-18-crown-6.
법적 정보
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 1
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
이미 열람한 고객
Growth and dissolution kinetics of potassium sulfate crystals in an agitated vessel.
Garside J, et al.
Industrial & Engineering Chemistry Fundamentals, 13(4), 299-305 (1974)
Thermodynamics of the vaporization processes for potassium sulfate.
Eliezer I and Howald RA.
J. Chem. Phys., 65(8), 3053-3062 (1976)
Sulfonation of benzocrown ethers by potassium sulfate in polyphosphoric acid.
Grebenyuk AD, et al.
Chemistry of Heterocyclic Compounds, 37(7), 822-826 (2001)
Rice bronzing in Nigeria caused by nutrient imbalances and its control by potassium sulfate application.
Yamauchi M.
Plant and Soil, 117(2), 275-286 (1989)
Conversion of phosphogypsum to potassium sulfate.
Aagli A, et al.
Journal of Thermal Analysis and Calorimetry, 82(2), 395-399 (2005)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.