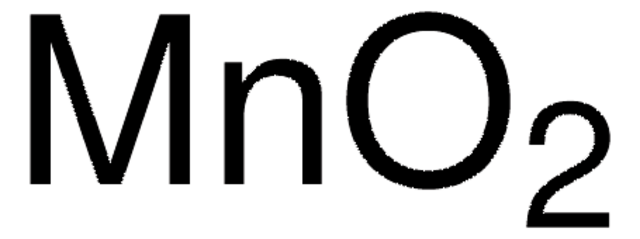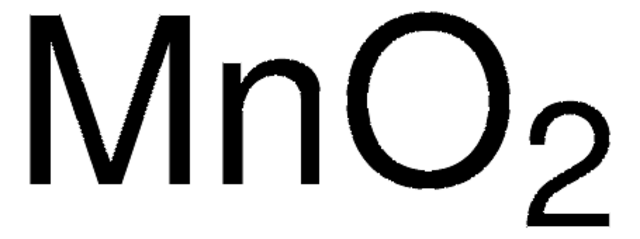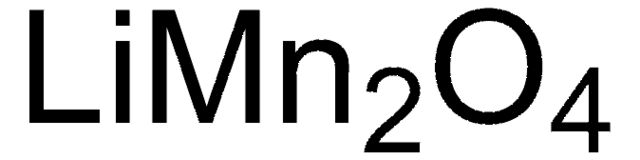추천 제품
Grade
reagent grade
분석
≥90%
양식
powder
입자 크기
10 μm
mp
535 °C (dec.) (lit.)
SMILES string
O=[Mn]=O
InChI
1S/Mn.2O
InChI key
NUJOXMJBOLGQSY-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Manganese(IV) oxide is an oxidizing reagent that can be used for the oxidation of propargylic alcohols, benzylic or heterocyclic alcohols, saturated alcohols, 1,2-diols, allylic alcohols to α, β-ethylenic aldehydes or ketones, and amines to aldehydes, imines, amides, and diazo compounds. It can also be used for the conversion of allylic alcohols to α, β-ethylenic esters or amides, hydration of nitriles to amides, dehydrogenation and aromatization reactions.
애플리케이션
- High-oxidation-state 3d metal complexes: Explores the catalytic properties of manganese(IV) oxide within high-oxidation-state complexes for advanced organic synthesis, demonstrating its critical role in accelerating chemical reactions and enhancing yield efficiencies, beneficial for pharmaceutical and chemical industries (Cheng J et al., 2018).
- Synthesis and properties of manganese complexes: Details the synthesis of new manganese complexes that demonstrate unique redox properties, useful for understanding electron transfer processes in various chemical and environmental contexts (Baffert C et al., 2002).
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - STOT RE 2 Inhalation
표적 기관
Brain
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 2
Flash Point (°F)
does not flash
Flash Point (°C)
does not flash
이미 열람한 고객
Manganese dioxide
Cahiez G, et al.
e-EROS Encyclopedia of Reagents for Organic Synthesis (2001)
Wen-Hui Kuan et al.
Journal of hazardous materials, 239-240, 152-159 (2012-09-25)
This study examined the reaction of methylene blue (MB) with tunneled manganese oxide pyrolusite regarding pH and reaction time. MB was cleaved through N-demethylation, in which reaction azure B (AB), azure A (AA), azure C (AC), and thionin (TH) were
Heng Lai et al.
ACS applied materials & interfaces, 4(5), 2325-2328 (2012-05-02)
MnO(2) nanoflakes coated on carbon nanohorns (CNHs) has been synthesized via a facile solution method and evaluated as anode for lithium-ion batteries. By using CNHs as buffer carrier, MnO(2)/CNH composite displays an excellent capacity of 565 mA h/g measured at
Xihong Lu et al.
Advanced materials (Deerfield Beach, Fla.), 24(7), 938-944 (2012-03-10)
WO3–x@Au@MnO2 core–shell nanowires (NWs) are synthesized on a flexible carbon fabric and show outstanding electrochemical performance in supercapacitors such as high specific capacitance, good cyclic stability, high energy density, and high power density. These results suggest that the WO3–x@Au@MnO2 NWs
Y Wang et al.
Journal of colloid and interface science, 380(1), 8-15 (2012-06-02)
Bio-inspired chemical approach has been developed for the surface modification and electrophoretic deposition of manganese dioxide and zirconia nanoparticles, prepared by chemical precipitation methods. Caffeic acid, trans-cinnamic acid, p-coumaric acid, and 2,4-dihydroxycinnamic acid were investigated for the surface modification of
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.