239267
Cobalt(II) nitrate hexahydrate
ACS reagent, ≥98%
동의어(들):
Cobaltous nitrate, Cobaltous nitrate hexahydrate, Nitric acid, cobalt(II) salt
About This Item
98.0-102.0%
powder, crystals or chunks
solid
추천 제품
Grade
ACS reagent
Quality Level
분석
≥98%
98.0-102.0%
양식
chips
powder, crystals or chunks
solid
반응 적합성
reagent type: catalyst
core: cobalt
불순물
≤0.01% insolubles
pH
4 (20 °C, 100 g/L)
mp
55 °C (lit.)
음이온 미량물
chloride (Cl-): ≤0.002%
sulfate (SO42-): ≤0.005%
양이온 미량물
Ca: ≤0.005%
Cu: ≤0.002%
Fe: ≤0.001%
K: ≤0.01%
Mg: ≤0.005%
NH4+: ≤0.2%
Na: ≤0.05%
Ni: ≤0.15%
Pb: ≤0.002%
Zn: ≤0.01%
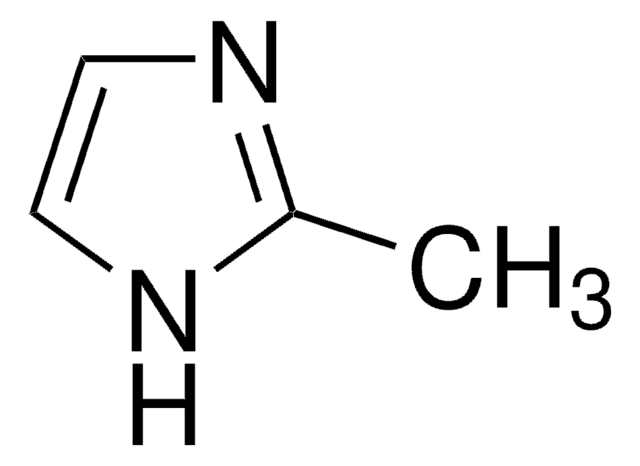
SMILES string
O.O.O.O.O.O.[Co++].[O-][N+]([O-])=O.[O-][N+]([O-])=O
InChI
1S/Co.2NO3.6H2O/c;2*2-1(3)4;;;;;;/h;;;6*1H2/q+2;2*-1;;;;;;
InChI key
QGUAJWGNOXCYJF-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
애플리케이션
- 5-Carboxanilide-dihydropyrimidinone derivatives by the condensation of acetoacetanilides, aldehydes, and urea/thiourea.
- Nitro indoles by the nitration of indoles using tert-butyl nitrite as the nitro source.
- Quinazolinones from 2-aminobenzonitriles using alcohols in the presence of phosphine ligand.
신호어
Danger
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 1B Inhalation - Eye Dam. 1 - Muta. 2 - Ox. Sol. 2 - Repr. 1B - Resp. Sens. 1 - Skin Sens. 1 - STOT RE 2 Inhalation
표적 기관
Lungs
Storage Class Code
5.1B - Oxidizing hazardous materials
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
이미 열람한 고객
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.