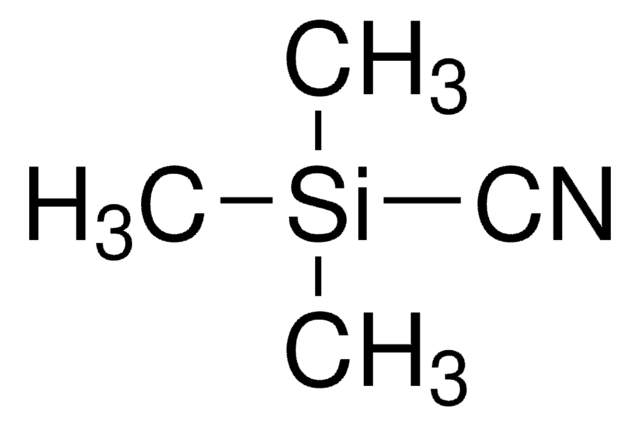
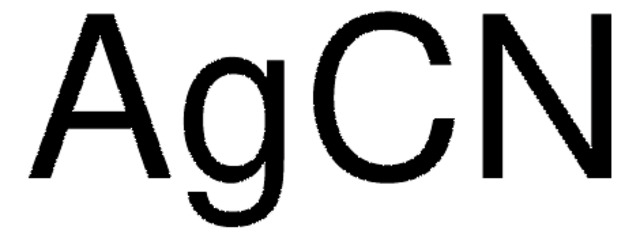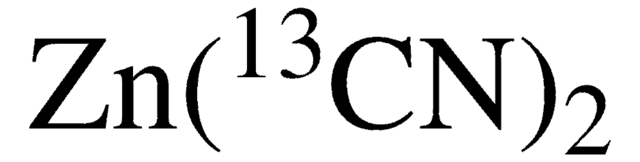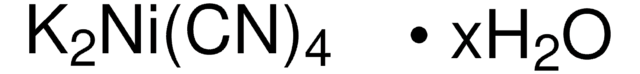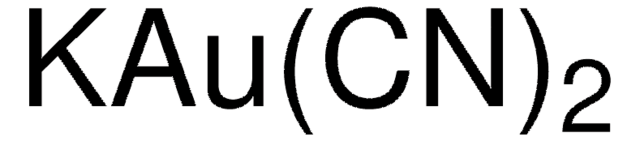추천 제품
분석
99%
형태
powder
mp
474 °C (lit.)
density
2.92 g/mL at 25 °C (lit.)
SMILES string
[Cu]C#N
InChI
1S/CN.Cu/c1-2;
InChI key
DULSAGLWMRMKCQ-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
애플리케이션
Applied in a widely useful, thiosulfate-assisted, synthesis of a variety of diamine-CuCN complexes.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 1 Dermal - Acute Tox. 2 Inhalation - Acute Tox. 2 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - STOT RE 2 Oral
표적 기관
Liver,spleen,Bone marrow
보충제 위험성
Storage Class Code
6.1B - Non-combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
Douglas B Grotjahn et al.
Journal of the American Chemical Society, 124(20), 5895-5901 (2002-05-16)
Copper(I) cyanide is an important reagent in organic, organometallic, and supramolecular chemistry because of both the copper center and the versatile cyanide ligand. Solid-phase CuCN and many of its derivatives show oligomeric or polymeric structures, a trait shared by other
Virender K Sharma et al.
Environmental science & technology, 39(10), 3849-3854 (2005-06-15)
Copper(Il) cyanide (Cu(CN)4(3-)) in the gold mine industry presentsthe biggest concern in cyanide management because it is much more stable than free cyanide. Cu(CN)4(3-) is highlytoxic to aquatic life; therefore, environmentally friendly techniques are required for the removal of Cu(CN)4(3-)
Fred B. Stocker et al.
Inorganic chemistry, 35(11), 3145-3153 (1996-05-22)
The syntheses and crystal structures of the first cyanide, sulfur mixed ligand copper(I) complexes are reported. The first complex of the family was discovered when (CuCN)(3)(C(6)H(12)N(4))(2) (1) (C(6)H(12)N(4) = hexamethylenetetramine) was treated with aqueous thiourea. The sulfur ligands include thiourea
Crystal Structures of a Series of Complexes Produced by Reaction of Copper(I) Cyanide with Diamines.
Fred B. Stocker et al.
Inorganic chemistry, 38(5), 984-991 (2001-10-24)
A new synthetic procedure developed recently in our laboratories has made possible the synthesis of variety of new complexes of CuCN with diamines. Synthesis was effected by adding the ligand to a solution of CuCN in aqueous sodium thiosulfate. This
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.