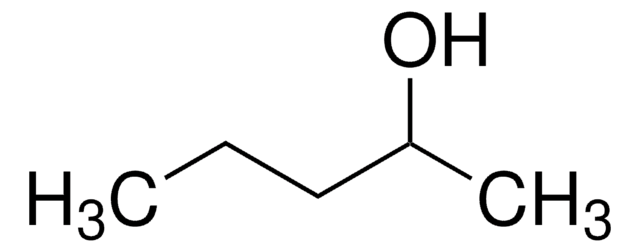
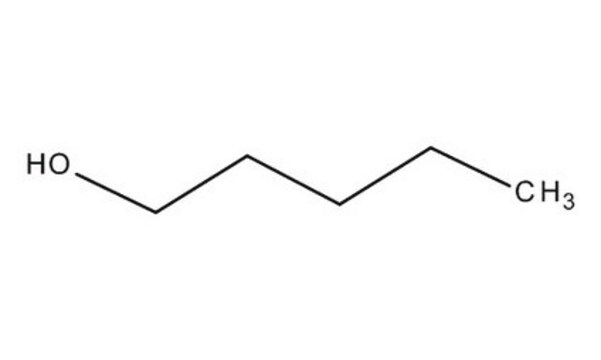
추천 제품
vapor density
3 (vs air)
Quality Level
vapor pressure
1 mmHg ( 13.6 °C)
1.5 mmHg ( 20 °C)
제품 라인
ReagentPlus®
분석
≥99%
형태
liquid
autoignition temp.
572 °F
expl. lim.
10 %, 100 °F
refractive index
n20/D 1.409 (lit.)
pH
7
bp
136-138 °C (lit.)
mp
−78 °C (lit.)
density
0.811 g/mL at 25 °C (lit.)
SMILES string
CCCCCO
InChI
1S/C5H12O/c1-2-3-4-5-6/h6H,2-5H2,1H3
InChI key
AMQJEAYHLZJPGS-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
1-Pentanol is a linear alcohol. Pentane, a ″next generation″ alcohol fuel, is a promising substitute for conventional gasoline and diesel fuels. Kinetics of thermal decomposition of its isomeric forms (1-pentanol, 2-methyl-1-butanol, and 3-methyl-1-butanol) have been reported.
1-Pentanol is a colorless liquid having a pleasant odor, which can be obtained as a product from the process of fractional distillation of mixed alcohols resulting from the chlorination and alkaline hydrolysis of pentane occurs.
1-Pentanol is a colorless liquid having a pleasant odor, which can be obtained as a product from the process of fractional distillation of mixed alcohols resulting from the chlorination and alkaline hydrolysis of pentane occurs.
애플리케이션
1-Pentanol may be used as a cosurfactant in the water-in-oil mixed surfactant microemulsions.
법적 정보
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Inhalation - Eye Dam. 1 - Flam. Liq. 3 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
3 - Flammable liquids
WGK
WGK 1
Flash Point (°F)
120.2 °F - closed cup
Flash Point (°C)
49 °C - closed cup
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
Harbison.DR, et al.
Hamilton and Hardy's Industrial Toxicology, (6) (2015)
Long Zhao et al.
The journal of physical chemistry. A, 116(37), 9238-9244 (2012-08-23)
Pentanol is one of the promising "next generation" alcohol fuels with high energy density and low hygroscopicity. In the present work, dominant reaction channels of thermal decomposition of three isomers of pentanol: 1-pentanol, 2-methyl-1-butanol, and 3-methyl-1-butanol were investigated by CBS-QB3
Experimental and detailed kinetic modeling study of 1-pentanol oxidation in a JSR and combustion in a bomb.
Togbe C, et al.
Proceedings of the Combustion Institute, 33(1), 367-374 (2011)
Anthony F Cann et al.
Applied microbiology and biotechnology, 85(4), 893-899 (2009-10-28)
Pentanol isomers such as 2-methyl-1-butanol and 3-methyl-1-butanol are a useful class of chemicals with a potential application as biofuels. They are found as natural by-products of microbial fermentations from amino acid substrates. However, the production titer and yield of the
Acid/base- and anion-controllable organogels formed from a urea-based molecular switch.
Sheng-Yao Hsueh et al.
Angewandte Chemie (International ed. in English), 49(48), 9170-9173 (2010-10-19)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.