Y0001400
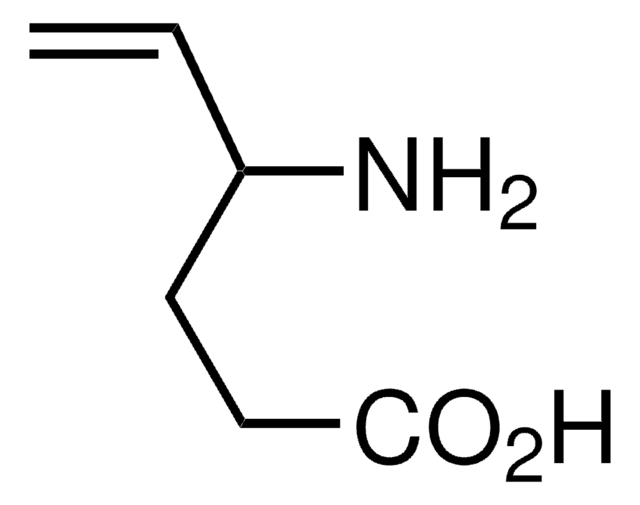
Vigabatrin
European Pharmacopoeia (EP) Reference Standard
동의어(들):
(±)-Vigabatrin, (R,S)-4-Amino-5-hexenoic acid, (±)-γ-Vinyl-GABA, (±)-4-Aminohexenoic acid
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C6H11NO2
CAS Number:
Molecular Weight:
129.16
MDL number:
UNSPSC 코드:
41116107
PubChem Substance ID:
NACRES:
NA.24
추천 제품
일반 설명
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
애플리케이션
Vigabatrin EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
생화학적/생리학적 작용
Irreversible GABA transaminase inhibitor. Increases intracellular concentration of GABA in nerve endings; possesses antiepileptic activity.
포장
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
기타 정보
Sales restrictions may apply.
신호어
Danger
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
STOT RE 1
표적 기관
Eyes,Central nervous system
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 1
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
Danièle Bentué-Ferrer et al.
Therapie, 65(1), 23-27 (2010-03-09)
Vigabatrin is a second generation anticonvulsant drug available in France since 1995. It is an amino acid analogue of the GABA, marketed under the racemic form [R(-)/S(+)50/50], but only the S(+)-enantiomer is active. Neither the mechanism of action of vigabatrin
E Ben-Menachem
Acta neurologica Scandinavica. Supplementum, (192)(192), 5-15 (2011-11-09)
Discovered more than three decades ago, vigabatrin is approved in more than 50 countries as adjunctive therapy for adult patients with refractory complex partial seizures who have responded inadequately to several alternative treatments and as monotherapy for pediatric patients aged
Justin A Tolman et al.
Expert opinion on pharmacotherapy, 10(18), 3077-3089 (2009-12-04)
Vigabatrin (Sabril) was approved in the USA in mid-2009 for the adjunctive treatment of refractory complex partial seizures and as treatment of infantile spasms. Vigabatrin's more than 30-year history of research and development is condensed into a clinically relevant review
S D Walker et al.
Acta neurologica Scandinavica. Supplementum, (192)(192), 72-82 (2011-11-09)
Vigabatrin is an effective antiepileptic drug (AED) for the treatment of refractory complex partial seizures (rCPS) and infantile spasms (IS). In clinical trials, vigabatrin was generally well-tolerated with an adverse event profile similar to that of other AEDs. The most
J M Pellock
Acta neurologica Scandinavica. Supplementum, (192)(192), 83-91 (2011-11-09)
Vigabatrin is an effective and well-tolerated antiepileptic drug (AED) for the treatment of refractory complex partial seizures (rCPS) and infantile spasms (IS), but its benefits must be evaluated in conjunction with its risk of retinopathy with the development of peripheral
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.