SMB01361
Phosphoramide mustard cyclohexylamine
≥95% (HPLC)
동의어(들):
N,N-Bis(2-Chloroethyl)phosphorodiamidic acid, cyclohexylamine, Friedman acid, cyclohexylamine, PAM cyclohexylamine, PM cyclohexylamine, PMC, Phosphamide mustard, Cyclohexylamine, Phosphorodiamidic mustard, Cyclohexylamine
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
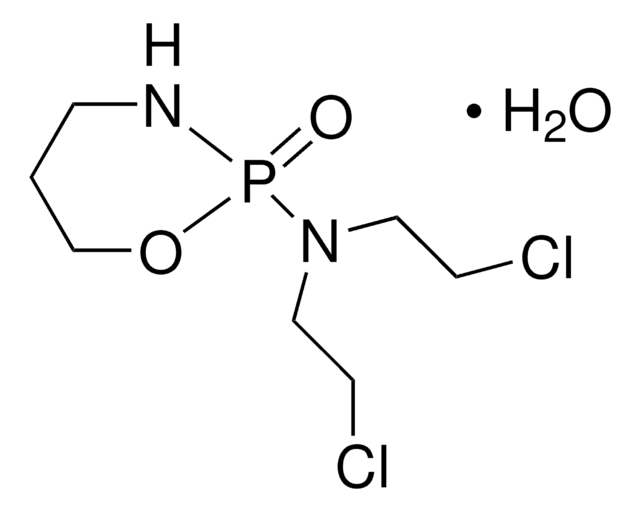
Linear Formula:
C4H11Cl2N2O2P · C6H13N
CAS Number:
MDL number:
UNSPSC 코드:
12352209
NACRES:
NA.22
추천 제품
Quality Level
분석
≥95% (HPLC)
양식
solid
저장 온도
-10 to -25°C
SMILES string
ClCCN(P(N)(O)=O)CCCl.NC1CCCCC1
InChI
1S/C6H13N.C4H11Cl2N2O2P/c7-6-4-2-1-3-5-6;5-1-3-8(4-2-6)11(7,9)10/h6H,1-5,7H2;1-4H2,(H3,7,9,10)
InChI key
BGTIPRUDEMNRIP-UHFFFAOYSA-N
일반 설명
Phosphoramide mustard cyclohexanamine, a potent cytotoxic metabolite derived from the anticancer drug cyclophosphamide, acts as a remarkable alkylating agent. It is generated from cyclophosphamide through the ring-opened tautomer of the cytochrome P450 (CYP) isoform-formed intermediate 4-hydroxycyclophosphamide. This metabolite exerts its cytotoxic effects by inducing DNA crosslinking, guanine alkylation, and the formation of DNA-protein conjugates. In a concentration-dependent manner, it specifically targets HT-1080 human fibrosarcoma cells, triggering DNA damage. An essential contributor to Cyclophosphamide′s anticancer activity, Phosphoramide Mustard Cyclohexanamine exhibits great promise for cancer research, as it has the potential to induce DNA adduct formation and activate the ovarian DNA repair response in granulosa cells.
애플리케이션
Used in the development of chemotherapeutic drugs that are known to treat cancer.
생화학적/생리학적 작용
Phosphoramide mustard cyclohexanamine induces cytotoxicity by forming cross-linked DNA adducts that hinder DNA strand separation during replication. This compound destroys rapidly dividing cells and leads to DNA damage. In a study with rat ovarian granulosa cells (SIGCs), it reduced cell viability and increased DNA damage response (DDR) gene expression and protein levels. In animal models, it exhibited significant anticancer activity by inhibiting tumor growth. These findings highlight the potential of Phosphoramide mustard cyclohexanamine in cancer research
특징 및 장점
- High quality compound suitable for multiple research applications
- Compatible with a wide variety of chromatographic and spectrometry techniques
기타 정보
For additional information on our range of Biochemicals, please complete this form.
This product is intended for research purposes only, and it is not meant for human consumption.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
Kendra L Clark et al.
Biology of reproduction, 102(1), 248-260 (2019-08-23)
Ataxia-telangiectasia-mutated (ATM) protein recognizes and repairs DNA double strand breaks through activation of cell cycle checkpoints and DNA repair proteins. Atm gene mutations increase female reproductive cancer risk. Phosphoramide mustard (PM) induces ovarian DNA damage and destroys primordial follicles, and
S A M Gernaat et al.
Scientific reports, 11(1), 2707-2707 (2021-02-03)
Cyclophosphamide (CPA) dosing by body surface area (BSA, m2) has been questioned as a predictor for individual drug exposure. This study investigated phosphoramide mustard-hemoglobin (PAM-Hb, pmol g-1 Hb) as a biomarker of CPA exposure in 135 female breast cancer patients
Julia Delahousse et al.
Journal for immunotherapy of cancer, 8(2) (2020-08-14)
Oxazaphosphorines (cyclophosphamide (CPA), ifosfamide (IFO)) are major alkylating agents of polychemotherapy protocols but limiting their toxicity and increasing their efficacy could be of major interest. Oxazaphosphorines are prodrugs that require an activation by cytochrome P450 (CYP). CPA is mainly metabolized
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.