추천 제품
product name
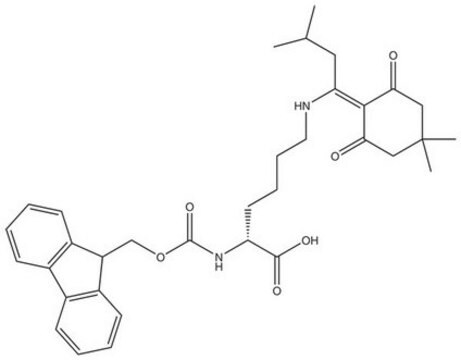
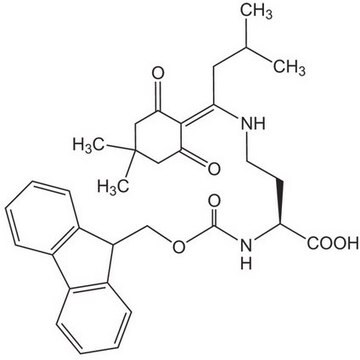
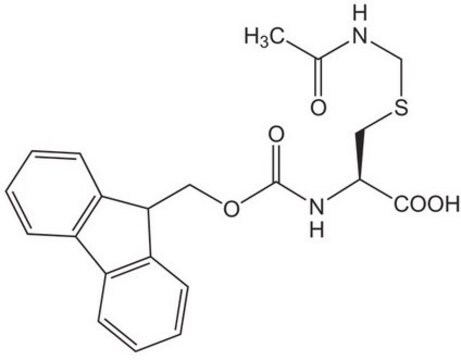
Fmoc-Dpr(ivDde)-OH, Novabiochem®
Quality Level
제품 라인
Novabiochem®
분석
≥95.0% (acidimetric)
≥98% (TLC)
≥99.0% (HPLC)
형태
powder
반응 적합성
reaction type: Fmoc solid-phase peptide synthesis
제조업체/상표
Novabiochem®
응용 분야
peptide synthesis
작용기
amine
저장 온도
15-25°C
InChI
1S/C31H36N2O6/c1-18(2)13-24(28-26(34)14-31(3,4)15-27(28)35)32-16-25(29(36)37)33-30(38)39-17-23-21-11-7-5-9-19(21)20-10-6-8-12-22(20)23/h5-12,18,23,25,32H,13-17H2,1-4H3,(H,33,38)(H,36,37)/t25-/m0/s1
InChI key
HLIFXCXTXPXGNH-VWLOTQADSA-N
일반 설명
Associated Protocols and Technical Articles
Cleavage and Deprotection Protocols for Fmoc SPPS
Literature references
[1] S. R. Chhabra, et al. (1998) Tetrahedron Lett., 39, 1603.
[2] R. Wilhelm, et al., Poster 34 presented at the 16th American Peptide Symposium, Minneapolis, 1999.
[3] J. Beythien & P. Schneeberger, in ′Peptides 2000, Proc. 26th European Peptide Symposium′, EDK, Paris, 2001, pp. 361.
[4] B. Rohwedder, et al. (1998) Tetrahedron Lett., 39, 1175.
결합
분석 메모
Appearance of substance (visual): powder
Identity (IR): passes test
Enantiomeric purity: ≥ 99.5 % (a/a)
Purity (TLC(157A)): ≥ 98 %
Purity (TLC(CMA2)): ≥ 98 %
Assay (HPLC, area%): ≥ 99.0 % (a/a)
Solubility (1 mmole in 2 ml DMF): clearly soluble
Assay (acidimetric): ≥ 95.0 %
Water (K. F.): ≤ 1.00 %
To see the solvent systems used for TLC of Novabiochem® products please click here.
법적 정보
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
문서
Novabiochem® product range has one of the largest collections of orthogonally and quasi-orthogonally protected tri-functional amino acids. These derivatives are useful tools for the synthesis of cyclic and branched peptides and peptides carrying side-chain modifications.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.