추천 제품
형태
powder
포장
pkg of 1 × 5 mg (999995P-5mg)
제조업체/상표
Avanti Research™ - A Croda Brand 999995P
지질 유형
cardiolipins
phospholipids
배송 상태
dry ice
저장 온도
−20°C
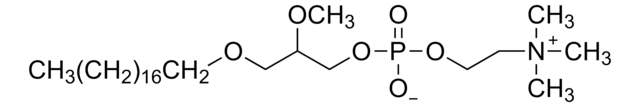
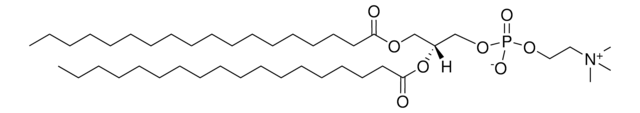
SMILES string
[O-]P(OCC[N+](C)(C)C)(OC[C@]([H])(OC)COCCCCCCCCCCCCCCCCCC)=O
InChI
1S/C10H24NO5P/c1-6-10(14-5)9-16-17(12,13)15-8-7-11(2,3)4/h10H,6-9H2,1-5H3/t10-/m0/s1
InChI key
GVMCXWJIRSIWFJ-JTQLQIEISA-N
애플리케이션
Edelfosine or 1-O-octadecyl-2-O-methyl-sn-glycero-3-phosphocholine might be used:
- as a non-hydrolysable LysoPC (phospholipid) analog for analyzing its ability to block sexual commitment in Plasmodium falciparum
- in multilamellar vesicle preparation, to study its effect on model membranes
- in the selection and screening of mutagenized cells, having the ability to inhibit the transport of alkylphosphocholine drugs across the plasma membrane
생화학적/생리학적 작용
Edelfosine acts as a precursor for alkyl-lysophospholipids. It possesses apoptotic action against several cancer cells such as prostate, leukemia, brain and lung tumors. It is associated with cellular transport system, signaling transducing systems, cytokine synthesis and lipid metabolism. Edelfosine is known to control intracellular calcium levels. It is not mutagenic and its anti-tumor action requires its incorporation into the cell. Edelfosine is also found to block the replication of human immunodeficiency virus type I (HIV-I).
포장
5 mL Clear Glass Sealed Ampule (999995P-5mg)
법적 정보
Avanti Research is a trademark of Avanti Polar Lipids, LLC
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
Alessio Ausili et al.
The journal of physical chemistry. B, 112(37), 11643-11654 (2008-08-21)
The effect of edelfosine (1- O-octadecyl-2- O-methyl-rac-glycero-3-phosphocholine or ET-18-OCH3) on model membranes containing 1-palmitoyl-2-oleoyl- sn-glycero-3-phosphocholine/sphingomyelin/cholesterol (POPC/SM/cholesterol) was studied by several physical techniques. The sample POPC/SM (1:1 molar ratio) showed a broad phase transition as seen by DSC, X-ray diffraction, and
Pamela K Hanson et al.
The Journal of biological chemistry, 278(38), 36041-36050 (2003-07-05)
The alkylphosphocholine class of drugs, including edelfosine and miltefosine, has recently shown promise in the treatment of protozoal and fungal diseases, most notably, leishmaniasis. One of the major barriers to successful treatment of these infections is the development of drug
Bruno M Castro et al.
The journal of physical chemistry. B, 117(26), 7929-7940 (2013-06-07)
Edelfosine (1-O-octadecyl-2-O-methyl-sn-glycero-phosphocholine) and miltefosine (hexadecylphosphocholine) are synthetic alkylphospholipids (ALPs) that are reported to selectively accumulate in tumor cell membranes, inducing Fas clustering and activation on lipid rafts, triggering apoptosis. However, the exact mechanism by which these lipids elicit these events
Alessio Ausili et al.
Langmuir : the ACS journal of surfaces and colloids, 34(28), 8333-8346 (2018-06-21)
Edelfosine is an anticancer drug with an asymmetric structure because, being a derivative of glycerol, it possesses two hydrophobic substituents of very different lengths. We showed that edelfosine destabilizes liquid-ordered membranes formed by either 1-palmitoyl-2-oleoyl- sn-glycero-3-phosphocholine, sphingomyelin (SM), and cholesterol
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.