추천 제품
분석
≥95%
Quality Level
양식
powder
분자량
229.32
반응 적합성
reagent type: cross-linking reagent
저장 조건
desiccated
solubility
DMSO or DMF: soluble
작용기
hydrazide
배송 상태
ambient
저장 온도
2-8°C
SMILES string
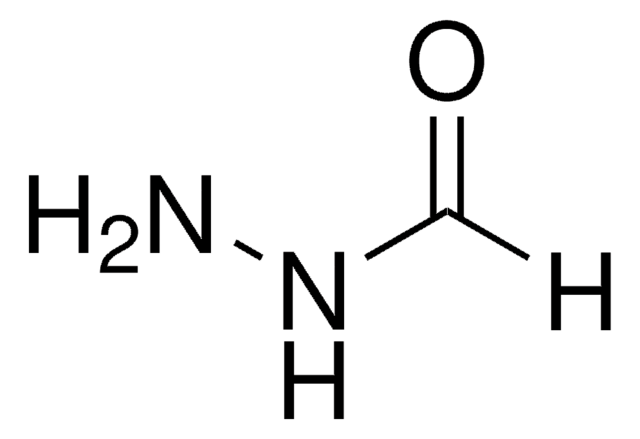
NNC(CCSSC1=NC=CC=C1)=O
InChI
1S/C8H11N3OS2/c9-11-7(12)4-6-13-14-8-3-1-2-5-10-8/h1-3,5H,4,6,9H2,(H,11,12)
InChI key
NITXODYAMWZEJY-UHFFFAOYSA-N
관련 카테고리
일반 설명
The PDPH is a heterobifunctional crosslinker containing sulfhydryl-reactive pyridyldithiol and carbonyl-reactive hydrazide moieties. Pyridyldithiols react with free sulfhydryls (-SH) to form disulfide bonds. Hydrazide groups react with carbonyls (aldehydes and ketones) to form stable hydrazone bonds. Aldehyde groups can be created by periodate-oxidation of sialic acid and other sugar components of glycoprotein polysaccharides. Thus, PDPH is useful for conjugating glycoproteins and sulfhydryl-containing peptides or proteins. Likewise, PDPH is useful as a sulfhydryl-addition reagent for glycoproteins and other carbohydrates; after reaction of the hydrazide to an oxidized carbohydrate, the pyridyldithiol group can be cleaved by a reducing agent to expose a sulfhydryl group. Yet another application for PDPH is to react the primary amine of the hydrazide moiety to a carboxyl group using the crosslinker EDC.
특징 및 장점
- Reactive groups: pyridyldisulfide and hydrazide
- Reactive toward: sulfhydryl groups and carbonyl (aldehyde) groups
- Short (9.2A), sulfhydryl-to-aldehyde crosslinker with disulfide bond spacer arm (cleavable)
- Pyridyldithiol group results in attachment to sulfhydryls via disulfide bond, which can be cleaved with DTT, TCEP or other reducing agents
- Hydrazide group conjugates to oxidized sugars of glycoproteins and carbohydrates
- Use sodium meta-periodate to oxidize glycosylation (e.g., sialic acid) to reactive aldehyde groups
- Use with EDC to conjugate primary amine of hydrazide group to carboxyl groups
주의사항
This product is sensitive to moisture. The vial is packaged in a resealable bag with a desiccant to reduce exposure to moisture. After cold storage, equilibrate the vial to room temperature before opening to reduce condensation inside the vial. Make fresh solutions. Storage of stock solutions is not recommended. After use, return the vial to the resealable bag. Close the bag and store the product at the recommended temperature.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
D Pain et al.
Journal of immunological methods, 40(2), 219-230 (1981-01-01)
Protein A-peroxidase monoconjugate was prepared in solution using a heterobifunctional reagent, N-succinimidyl-3-(2-pyridyldithio)-propionate. The yield of the monoconjugate was much higher than that obtained with current methods. An immunoassay method was developed in which protein A-peroxidase monoconjugate served as a universal
G N Ranadive et al.
Nuclear medicine and biology, 20(6), 719-726 (1993-08-01)
We have developed a very efficient labeling technique for monoclonal antibodies with technetium-99m. Oxidation of sugar residues on the IgG class of antibodies leads to the generation of aldehyde groups which are further reacted with two newly developed hydrazide compounds.
D J O'Shannessy et al.
Immunology letters, 8(5), 273-277 (1984-01-01)
A novel method is described for the biotinylation of immunoglobulins. The procedure relies on the generation of reactive aldehydes on the carbohydrate moieties of the immunoglobulin by oxidation with sodium periodate and subsequent reaction with biotin hydrazide. The method is
Immobilization of glycoconjugates by their oligosaccharides: use of hydrazido-derivatized matrices.
D J O'Shannessy et al.
Analytical biochemistry, 191(1), 1-8 (1990-11-15)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.
![LC-SPDP (succinimidyl 6-[3(2-pyridyldithio)propionamido]hexanoate)](/deepweb/assets/sigmaaldrich/product/structures/300/586/d95fd80c-e201-4b0b-8aee-31e109c2ff41/640/d95fd80c-e201-4b0b-8aee-31e109c2ff41.png)

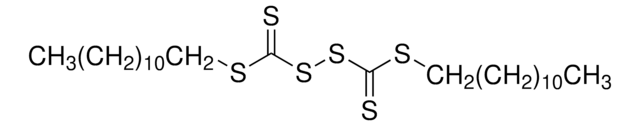
![Sulfo-LC-SPDP (sulfosuccinimidyl 6-[3′-(2-pyridyldithio)propionamido]hexanoate)](/deepweb/assets/sigmaaldrich/product/structures/266/633/e2a263be-4bd3-4fcf-89c4-75b5e2bd829c/640/e2a263be-4bd3-4fcf-89c4-75b5e2bd829c.png)