776181
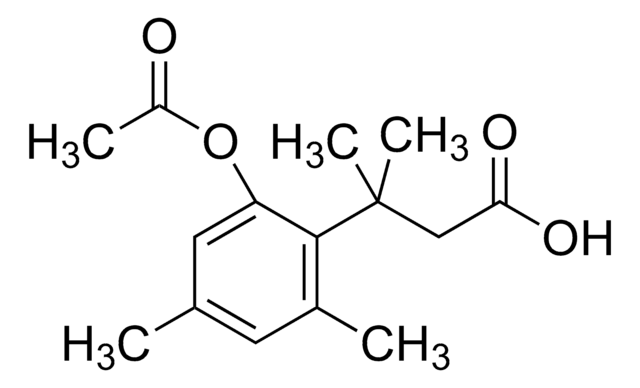
3-(2-Acetoxy-4,6-dimethylphenyl)-3-methylbutyric acid N-succinimidyl ester
95%
동의어(들):
3-(2-Acetoxy-4,6-dimethylphenyl)-3-methylbutyric acid N-hydroxysuccinimide ester, Trimethyl lock NHS, Trimethyl lock succinimidyl ester
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C19H23NO6
Molecular Weight:
361.39
MDL number:
UNSPSC 코드:
12352106
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
95%
형태
solid
mp
107-112 °C
저장 온도
2-8°C
SMILES string
CC(=O)Oc1cc(C)cc(C)c1C(C)(C)CC(=O)ON2C(=O)CCC2=O
InChI
1S/C19H23NO6/c1-11-8-12(2)18(14(9-11)25-13(3)21)19(4,5)10-17(24)26-20-15(22)6-7-16(20)23/h8-9H,6-7,10H2,1-5H3
InChI key
KMNOKEFBKJGRFL-UHFFFAOYSA-N
관련 카테고리
애플리케이션
This ′trimethyl lock′ derivative is activated with a succinimidyl ester for modification of amine containing compounds and biomolecules. After modification of your amine of interest, your now ′masked′ molecule can be released by cleavage of the ester, which can be achieved by cellular esterases. This triggers a rapid lactonization reaction of the ′trimethyl lock′ to regenerate your original amine-containing molecule. This approach has been applied to release of fluorophores, peptides, nucleic acids as well as small molecule drugs.
가장 최신 버전 중 하나를 선택하세요:
Michael N Levine et al.
Chemical science, 3(8), 2412-2420 (2012-11-28)
The trimethyl lock is an o-hydroxydihydrocinnamic acid derivative in which unfavorable steric interactions between three pendant methyl groups encourage lactonization to form a hydrocoumarin. This reaction is extremely rapid, even when the electrophile is an amide and the leaving group
Michael N Levine et al.
Molecules (Basel, Switzerland), 13(2), 204-211 (2008-02-29)
p-Nitrophenyl acetate is the most commonly used substrate for detecting the catalytic activity of esterases, including those that activate prodrugs in human cells. This substrate is unstable in aqueous solution, limiting its utility. Here, a stable chromogenic substrate for esterases
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.