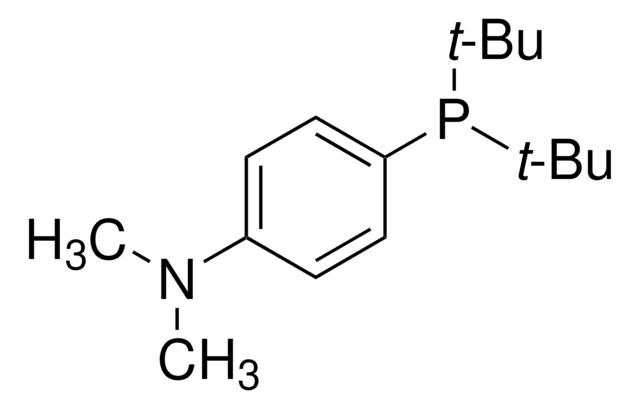
695459
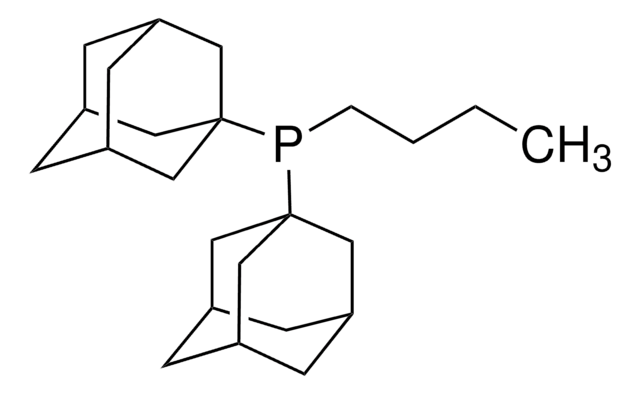
1,3,5,7-Tetramethyl-6-phenyl-2,4,8-trioxa-6-phosphaadamantane
97%
동의어(들):
1,3,5,7-Tetramethyl-8-phenyl-2,4,6-trioxa-8-phosphatricyclo[3.3.1.13,7]decane, meCgPPh
About This Item
추천 제품
Quality Level
분석
97%
형태
solid
반응 적합성
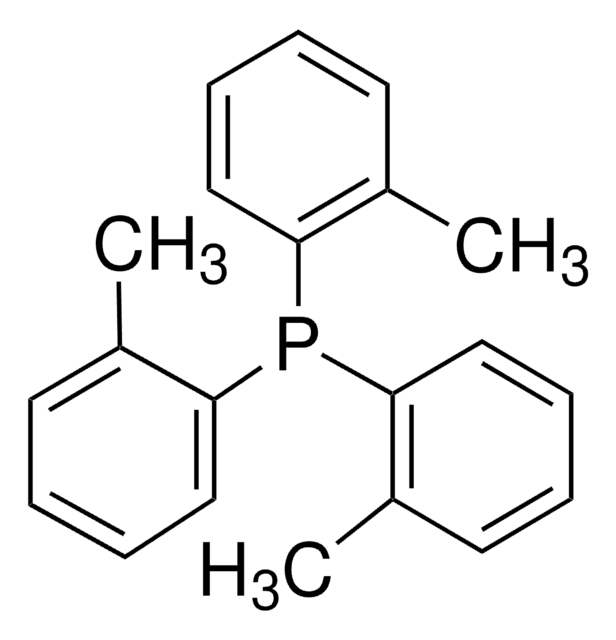
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: ligand
reaction type: Cross Couplings
mp
106-111 °C
작용기
phosphine
SMILES string
CC12CC3(C)OC(C)(CC(C)(O1)P3c4ccccc4)O2
InChI
1S/C16H21O3P/c1-13-10-15(3)19-14(2,17-13)11-16(4,18-13)20(15)12-8-6-5-7-9-12/h5-9H,10-11H2,1-4H3
InChI key
AVVSJWUWBATQBX-UHFFFAOYSA-N
애플리케이션
- As a ligand to synthesize complexes for hydroformylation catalysis.
- To catalyze the synthesis of dibenzo[b,f][1,4]oxazepin-11(10H)-ones and 3-methyl-3,4-dihydrocoumarins by intramolecular cyclocarbonylation.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
이미 열람한 고객
문서
The use of amines and phosphines in nucleophilic catalysis is well precedented; however, arguably one of the severe limitations with respect to exploiting the more nucleophilic, yet less basic, phosphine in this regard is its air sensitivity.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.![[(1,3,5,7-Tetramethyl-6-phenyl-2,4,6-trioxa-6-phosphaadamantane)-2-(2′-amino-1,1′-biphenyl)]palladium(II) methanesulfonate](/deepweb/assets/sigmaaldrich/product/structures/324/001/3ffb4bd2-9c6b-451c-80ee-a217f03ca932/640/3ffb4bd2-9c6b-451c-80ee-a217f03ca932.png)


![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)