659878
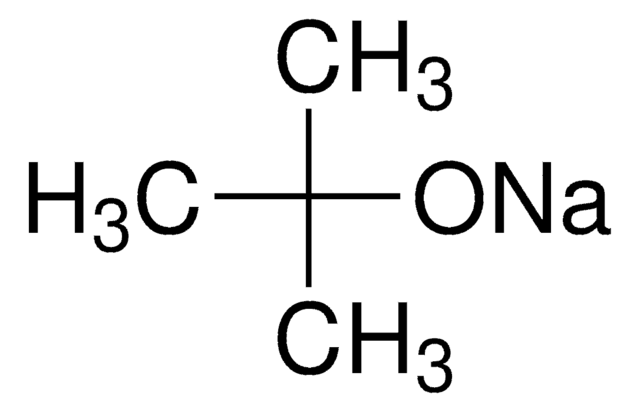
Potassium tert-butoxide
sublimed grade, 99.99% trace metals basis
동의어(들):
Potassium tert-butylate, Potassium t-butoxide
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
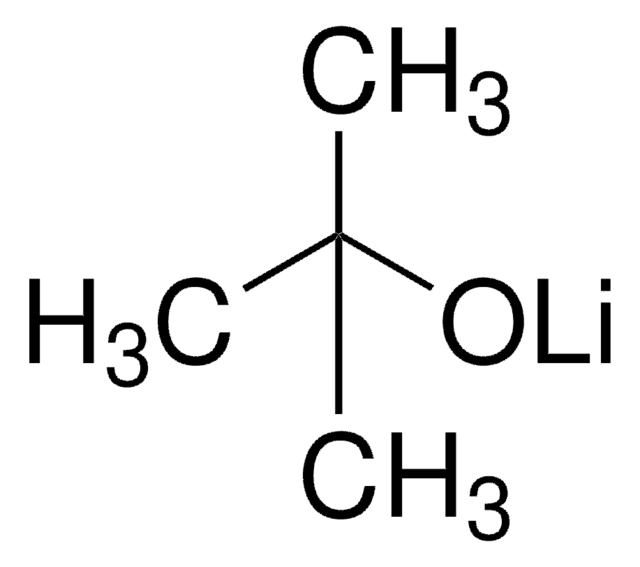
Linear Formula:
(CH3)3COK
CAS Number:
Molecular Weight:
112.21
Beilstein:
3556712
EC Number:
MDL number:
UNSPSC 코드:
12352103
PubChem Substance ID:
NACRES:
NA.23
추천 제품
Grade
sublimed grade
Quality Level
vapor pressure
1 mmHg ( 220 °C)
분석
99.99% trace metals basis
양식
solid
반응 적합성
core: potassium
mp
256-258 °C (dec.) (lit.)
SMILES string
[K+].CC(C)(C)[O-]
InChI
1S/C4H9O.K/c1-4(2,3)5;/h1-3H3;/q-1;+1
InChI key
LPNYRYFBWFDTMA-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Potassium tert-butoxide is a strong alkoxide base, it can deprotonate carbon and other Brφnsted acids. It is a relatively poor nucleophile.
Total impurities: may contain up to 5,000 ppm Sodium
애플리케이션
Mizoroki-Heck-Type reactions are mediated by potassium tert-butoxide. potassium tert-butoxide can commence the anionic polymerization of carbazolyl-substituted oxiranes.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Eye Dam. 1 - Flam. Sol. 1 - Self-heat. 2 - Skin Corr. 1A
보충제 위험성
Storage Class Code
4.2 - Pyrophoric and self-heating hazardous materials
WGK
WGK 1
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
이미 열람한 고객
Anionic polymerization of carbazolyl-substituted oxiranes initiated by potassium alkalide, potassium tert-butoxide and potassium hydride.
Buika G, et al.
Macromolecular Chemistry and Physics, 196(4), 1287-1293 (1995)
Einav Amit et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 26(57), 13046-13052 (2020-04-29)
N-heterocyclic carbenes (NHCs) have emerged as a unique molecular platform for the formation of self-assembled monolayers (SAMs) on various surfaces. However, active carbene formation requires deprotonation of imidazolium salt precursors, which is mostly facilitated by exposure of the salt to
Hajime Ito et al.
Chemical communications (Cambridge, England), 48(64), 8006-8008 (2012-07-10)
The regio- and diastereoselective silaboration of aromatic alkenes with a silylboron compound proceeds in the presence of a catalytic amount of potassium tert-butoxide, providing a complementary method to the corresponding transition metal-catalyzed reactions.
Aurélie Mallinger et al.
The Journal of organic chemistry, 74(3), 1124-1129 (2008-12-24)
3-Aryltetronic acids were prepared in one step by treatment of a mixture of methyl arylacetates and methyl hydroxyacetates with potassium tert-butoxide, via tandem transesterification/Dieckmann condensation. Several mushroom or lichen pigments, vulpinic acids, were synthesized from 3-(4-methoxyphenyl)tetronic acid in three steps
Scott E Denmark et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 12(19), 4954-4963 (2006-05-04)
This paper chronicles the conceptual development, proof of principle experiments, and recent advances in the palladium-catalyzed cross-coupling reactions of the conjugate bases of organosilanols. The discovery that led to the design and refinement of this process represents a classical illustration
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.