638439
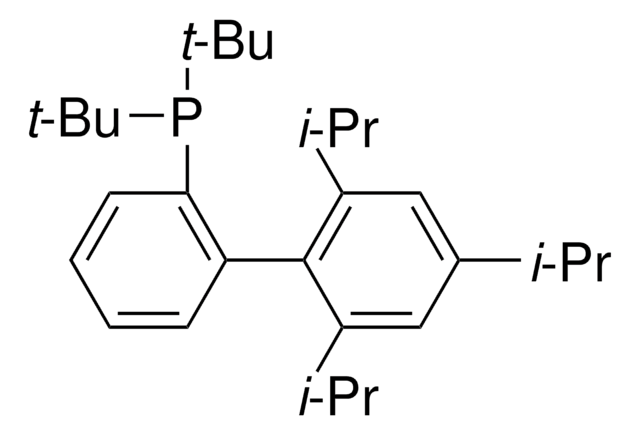
JohnPhos
97%
동의어(들):
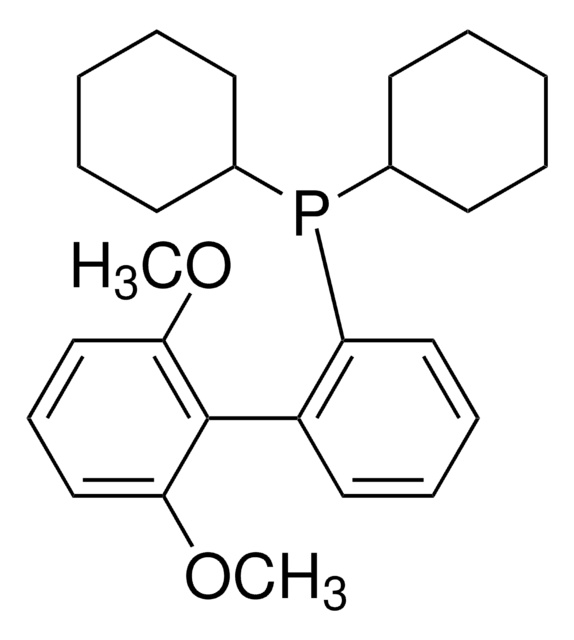
(2-Biphenyl)di-tert-butylphosphine, (2-Biphenylyl)di-tert-butylphosphine, 2-(Di-tert-butylphosphino)biphenyl
About This Item
추천 제품
Quality Level
분석
97%
반응 적합성
reaction type: Cross Couplings
reagent type: ligand
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reagent type: ligand
reaction type: C-X Bond Formation
reagent type: ligand
reaction type: Heck Reaction
reagent type: ligand
reaction type: Suzuki-Miyaura Coupling
mp
86-88 °C (lit.)
작용기
phosphine
SMILES string
CC(C)(C)P(c1ccccc1-c2ccccc2)C(C)(C)C
InChI
1S/C20H27P/c1-19(2,3)21(20(4,5)6)18-15-11-10-14-17(18)16-12-8-7-9-13-16/h7-15H,1-6H3
InChI key
CNXMDTWQWLGCPE-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Learn more about Buchwald Phosphine Ligands
애플리케이션
- Hydrophenoxylation of unactivated internal alkynes.
- Microwave-mediated Suzuki-Miyaura cross-coupling of benzylic bromides.
- Pharmaceutical synthesis of novel imidazo[1,2-a]pyridines, having potent activity against the herpes virus.
- Barluenga′s coupling of vinyl bromides with hydrazines.
- Pd-catalyzed 2,3-diarylation of α,α-disubstituted-3-thiophenemethanols, via cleavage of C-H and C-C bonds.
Catalyst for:
- Decarboxylative cross-coupling of dialkoxybenzoic acids with diaryl disulfides or diaryl diselenides
- Stereoselective preparation of imidazolidinones via intramolecular hydroamination of N-allylic-N-arylureas
- Regioselective arylation of olefins with aryl chlorides
- Cross-coupling reaction for the synthesis of polyunsaturated macrolactones
- Regioselective O-alkylation reactions
- Sonogashira-type cross coupling
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
이미 열람한 고객
문서
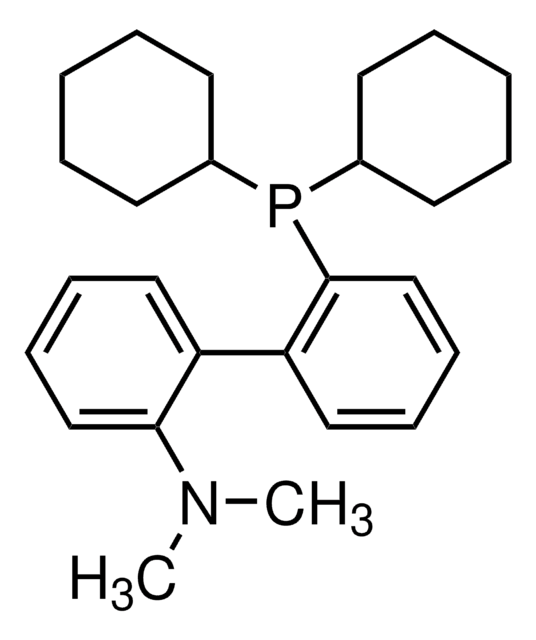
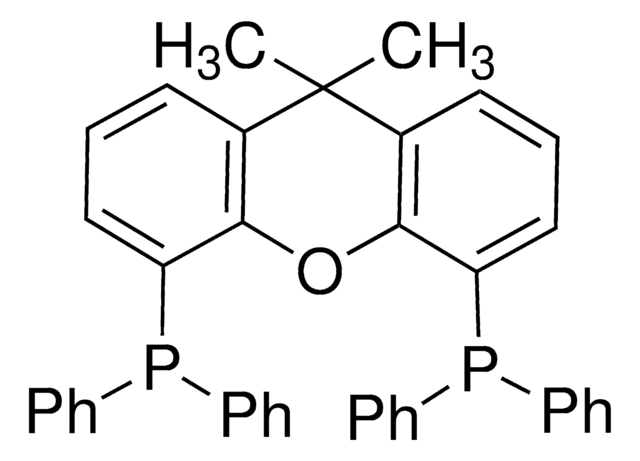
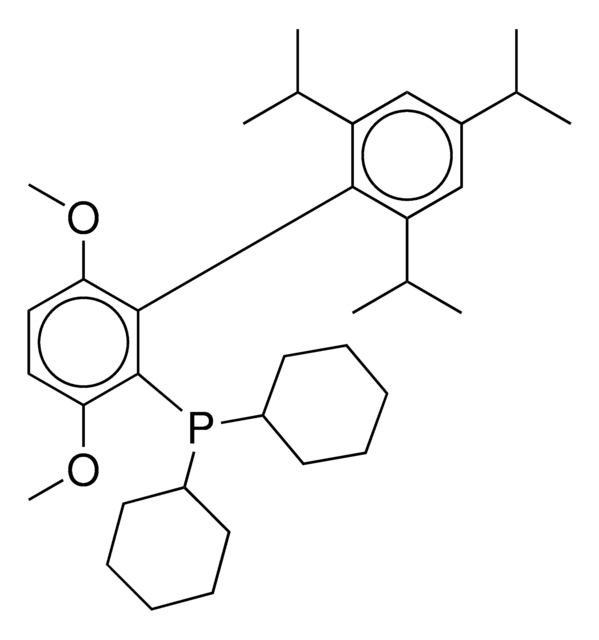
Buchwald Ligands
Buchwald Phosphine Ligands
Over the past several years, Pd-catalyzed cross-coupling of silicon compounds has rapidly gained acceptance as a suitable alternative to more commonly known methods such as: Stille (Sn), Kumada (Mg), Suzuki (B), and Negishi (Zn) cross-couplings.
Buchwald phosphine ligands for C-C, C-N, and C-O bond formation.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.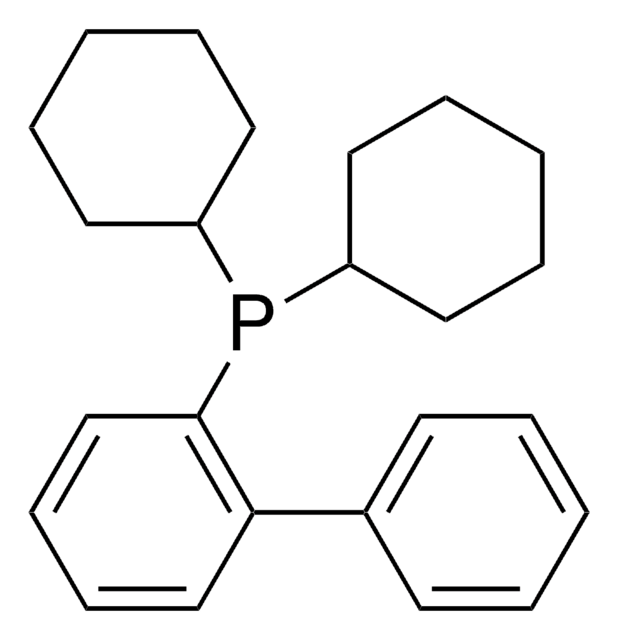


![Chloro[(1,1′-biphenyl-2-yl)di-tert-butylphosphine]gold(I) 98%](/deepweb/assets/sigmaaldrich/product/structures/192/300/6b6ed2fd-ac0b-4370-80e3-807dad65a825/640/6b6ed2fd-ac0b-4370-80e3-807dad65a825.png)