559210
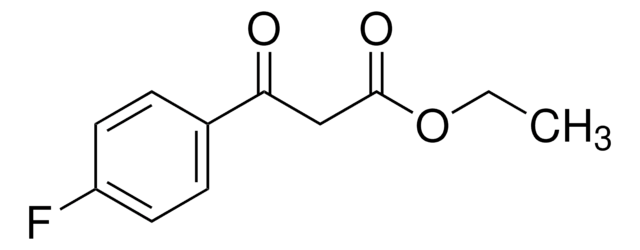
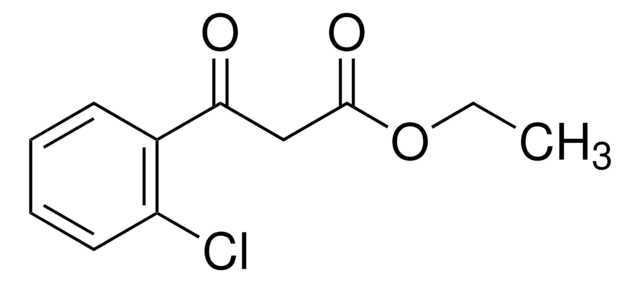
Ethyl (4-chlorobenzoyl)acetate
동의어(들):
3-(4-Chlorophenyl)-3-oxopropanoic acid ethyl ester, Ethyl 3-(4-chlorophenyl)-3-oxopropanoate, NSC 406743
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
Linear Formula:
ClC6H4COCH2CO2C2H5
CAS Number:
Molecular Weight:
226.66
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
양식
solid
refractive index
n20/D 1.5500 (lit.)
bp
268-269 °C (lit.)
density
1.218 g/mL at 25 °C (lit.)
작용기
chloro
ester
ketone
SMILES string
CCOC(=O)CC(=O)c1ccc(Cl)cc1
InChI
1S/C11H11ClO3/c1-2-15-11(14)7-10(13)8-3-5-9(12)6-4-8/h3-6H,2,7H2,1H3
InChI key
DGCZHKABHPDNCC-UHFFFAOYSA-N
애플리케이션
Ethyl (4-chlorobenzoyl)acetate may be used to synthesize 2-(carboethoxy)-3-(4′-chloro)phenylquinoxaline 1,4-dioxide.
Reactant involved in:
- Tandem condensation-cyclization reactions
- Stereoselective ketonization-olefination of indoles
- Synthesis of antiplasmodial agents
- SIRT 1/2 inhibitor preparation for use as antitumor agents
- Synthesis of mineralocorticoid receptor antagonists
- Intramolecular Michael addition reactions
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
Comparative Use of Solvent-free KF-Al2O3 and K2CO3 in Acetone in the Synthesis of Quinoxaline 1, 4-Dioxide Derivatives Designed as Antimalarial Drug Candidates.
Lima LM, et al.
Journal of Heterocyclic Chemistry, 42(7), 1381-1385 (2005)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.