추천 제품
분석
96%
refractive index
n20/D 1.603 (lit.)
bp
116-117 °C/12 mmHg (lit.)
density
1.484 g/mL at 25 °C (lit.)
저장 온도
2-8°C
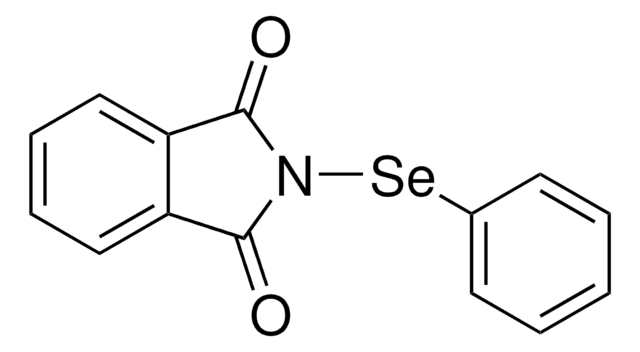
SMILES string
N#C[Se]c1ccccc1
InChI
1S/C7H5NSe/c8-6-9-7-4-2-1-3-5-7/h1-5H
InChI key
NODWRXQVQYOJGN-UHFFFAOYSA-N
일반 설명
Phenyl selenocyanate is a selenenylation agent that can be prepared by reacting benzeneselenenyl chloride and trimethylsilyl cyanide.
애플리케이션
Phenyl selenocyanate may be used in the synthesis of:
- benzeneselenol esters
- 2-bromoethyl phenyl selenie dibromide
- α,α-dioxy-β-phenylseleno carbonitriles
- β-alkoxyalkyl phenyl selenide
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - STOT RE 2
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
Flash Point (°F)
228.2 °F - closed cup
Flash Point (°C)
109 °C - closed cup
개인 보호 장비
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
이미 열람한 고객
Cyanoselenenylation of ketene acetals. Synthesis of carbonyl-protected a-oxo carbonitriles.
Tomoda S, et al.
Chemistry Letters (Jpn), 11(11), 1733-1734 (1982)
A convenient synthesis of phenyl selenocyanate.
Tomoda S, et al.
Chemistry Letters (Jpn), 10(8), 1069-1070 (1981)
Aryl selenocyanates and aryl thiocyanates: reagents for the preparation of activated esters.
Grieco PA, et al.
The Journal of Organic Chemistry, 43(6), 1283-1285 (1978)
Synthesis of Aryl 2-Haloethyl Selenides and their Reactions with Potassium Selenocyanate.
Lindgren B.
Acta Chemica Scandinavica. Series B, 31(1), 1-6 (1977)
Facile oxyselenation of olefins in the presence of copper (II) or copper (I) chloride as catalyst.
Toshimitsu A, et al.
The Journal of Organic Chemistry, 45(10), 1953-1958 (1980)
문서
For microbiologists the most fundamental stain was developed in 1884 by the Danish bacteriologist Hans Christian Gram.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.