모든 사진(1)
About This Item
실험식(Hill 표기법):
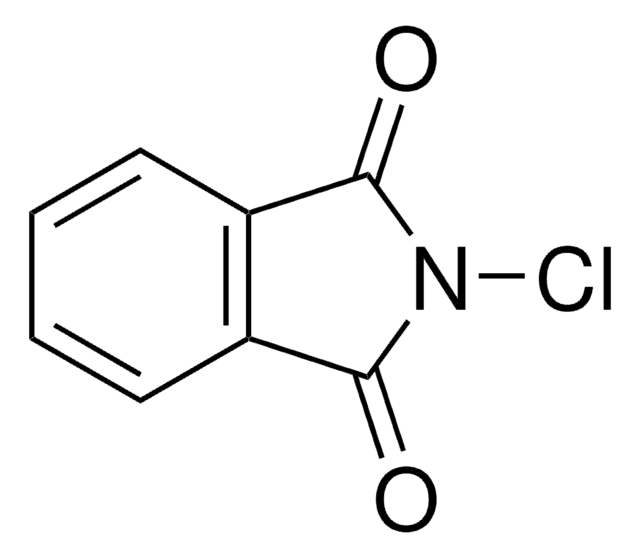
C7H4ClNO3S
CAS Number:
Molecular Weight:
217.63
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
분석
99%
mp
148-152 °C (lit.)
SMILES string
ClN1C(=O)c2ccccc2S1(=O)=O
InChI
1S/C7H4ClNO3S/c8-9-7(10)5-3-1-2-4-6(5)13(9,11)12/h1-4H
InChI key
VKWMGUNWDFIWNW-UHFFFAOYSA-N
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Oral - Carc. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
N Jayasree et al.
Journal - Association of Official Analytical Chemists, 70(4), 762-763 (1987-07-01)
Three simple titrimetric methods have been developed to determine iodine-bromine numbers of some edible oils, such as coconut, gingelly, groundnut, mustard, olive, palm olein, and sunflower, using 3 N-chloroimides. The 3 N-chloroimides are N-chlorophthalimide, N-chlorosuccinimide, and N-chlorosaccharin, all of which
Kevin I Booker-Milburn et al.
Organic letters, 5(18), 3313-3315 (2003-08-29)
[reaction: see text] N-Chlorosaccharin has been shown to undergo electrophilic Ritter-type reactions with alkenes in acetonitrile. The resulting labile beta-chloro sulfonylamidines can be ring-opened and cyclized to imidazolines. Overall this provides a one pot method for the electrophilic diamination of
N-chlorosaccharin as a possible chlorinating reagent: structure, chlorine potential, and stability in water and organic solvents.
H S Dawn et al.
Journal of pharmaceutical sciences, 59(7), 955-959 (1970-07-01)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.