추천 제품
Grade
technical grade
Quality Level
양식
viscous liquid
refractive index
n20/D 1.468 (lit.)
bp
133 °C/10 mmHg (lit.)
density
1.309 g/mL at 25 °C (lit.)
작용기
ester
hydroxyl
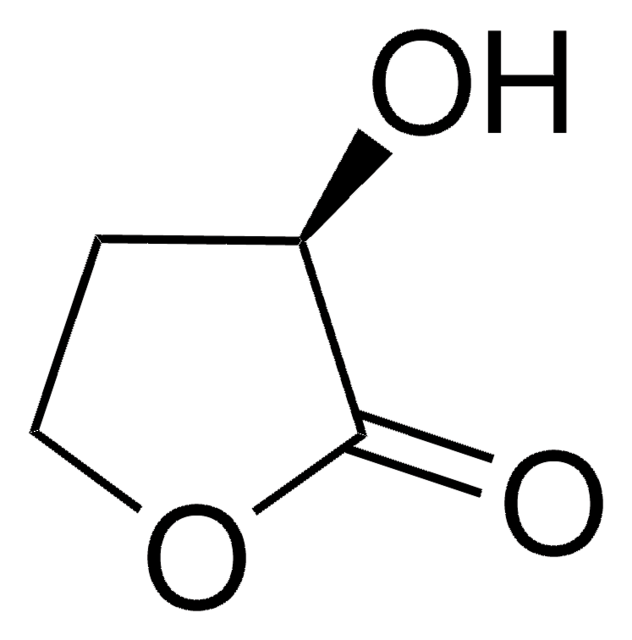
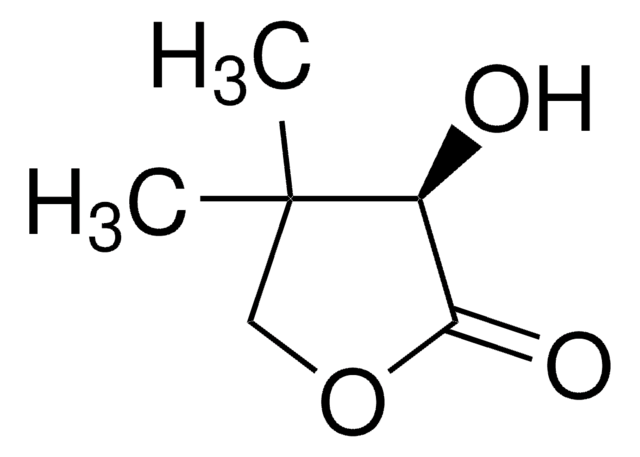
SMILES string
OC1CCOC1=O
InChI
1S/C4H6O3/c5-3-1-2-7-4(3)6/h3,5H,1-2H2
InChI key
FWIBCWKHNZBDLS-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
α-Hydroxy-γ-butyrolactone is a 5-membered cyclic ester. It was obtained via tin-conversion of biomass-derived 1,3-dihydroxyacetone (DHA) and formaldehyde.
애플리케이션
α-Hydroxy-γ-butyrolactone may be employed as starting reagent in the synthesis of series of seco-pseudonucleoside synthons via aminolysis. It may be employed as starting reagent in the synthesis of enantiomerically pure orthogonally protected δ-azaproline, via Mitsunobu reaction.
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point (°F)
235.4 °F - closed cup
Flash Point (°C)
113 °C - closed cup
개인 보호 장비
Eyeshields, Gloves, multi-purpose combination respirator cartridge (US)
이미 열람한 고객
Efficient synthesis of enantiomerically pure (S)-d-azaproline starting from (R)-a-hydroxy-?-butyrolactone via the Mitsunobu reaction.
Voss E, et al.
Tetrahedron Asymmetry, 20(15), 1809-1812 (2009)
Sho Yamaguchi et al.
Chemical communications (Cambridge, England), 50(35), 4600-4602 (2014-03-29)
The direct conversion of biomass-derived 1,3-dihydroxyacetone (DHA) and formaldehyde to α-hydroxy-γ-butyrolactone (HBL) was achieved through the use of tin(iv) chloride and a small amount of water and the yield reached up to 70%. The reaction mechanism was also investigated by
Natalia N Dioubankova et al.
Organic letters, 4(26), 4607-4610 (2002-12-20)
[reaction: see text] Two series of seco-pseudonucleoside synthons were synthesized from (R)-(+)-alpha-hydroxy-gamma-butyrolactone and (R)-(-)-pantolactone by aminolysis, side-chain protection, dimethoxytritylation, and phosphitylation or solid-phase attachment. The phosphoramidites and solid supports were used in automated DNA synthesis to prepare oligonucleotides modified with
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.