추천 제품
Quality Level
분석
97%
포함
Silver Wool as stabilizer
refractive index
n20/D 1.471 (lit.)
bp
140 °C/5 mmHg (lit.)
density
1.278 g/mL at 25 °C (lit.)
SMILES string
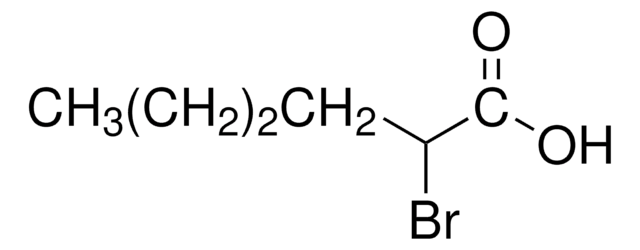
CCCCCCC(Br)C(O)=O
InChI
1S/C8H15BrO2/c1-2-3-4-5-6-7(9)8(10)11/h7H,2-6H2,1H3,(H,10,11)
InChI key
GTGTXZRPJHDASG-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
관련 카테고리
일반 설명
2-Bromooctanoic acid is an effective substitute for the expensive (2,000 times) and cell-growth-inhibiting polyhydroxyalkanoic acid synthesis inhibitor, cerulenin. It blocks the formation of polyhydroxyalkanoic acid in Pseudomonas fluorescens BM07 without any influence on the cell growth when grown on fructose. It inhibits β-oxidation of fatty acids in perfused rat liver and in mitochondria isolated from rat liver.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Skin Corr. 1B
Storage Class Code
8A - Combustible, corrosive hazardous materials
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
Channeling of intermediates derived from medium-chain fatty acids and de novo-synthesized fatty acids to polyhydroxyalkanoic acid by 2-bromooctanoic acid in Pseudomonas fluorescens BM07.
LEE H-J, et al.
Journal of microbiology and biotechnology, 14(6), 1256-1266 (2004)
B M Raaka et al.
The Journal of biological chemistry, 254(14), 6755-6762 (1979-07-25)
Incubation of rat liver mitochondria with 10 microM DL-2-bromooctanoate causes complete and irreversible inactivation of 3-ketothiolase I (acyl-CoA:acetyl-CoA C-acyltransferase). Evidence is presented that mitochondria convert bromooctanoate to 2-bromo-3-ketooctanoyl-CoA, an alpha-haloketone which is probably the active form of the inhibitor. The
W E Roediger et al.
British journal of experimental pathology, 67(6), 773-782 (1986-12-01)
There is some evidence that failure of fatty acid or beta-oxidation in the epithelium of the colonic mucosa is associated with the development of ulcerative colitis. We tested the hypothesis that inhibition of fatty acid oxidation in the colonic mucosa
N Mayorek et al.
The Journal of biological chemistry, 260(11), 6528-6532 (1985-06-10)
Triacylglycerol synthesis in cultured rat hepatocytes was inhibited by 2-bromooctanoate with a concomitant accumulation of diacylglycerols. 2-Bromooctanoate inhibition could be ascribed to its thioesterification by medium chain fatty acyl-CoA synthase (Raaka, B.M., and Lowenstein, J.M. (1979) J. Biol. Chem. 254
N Mayorek et al.
European journal of biochemistry, 182(2), 395-400 (1989-06-15)
The limiting role of diacylglycerol acyltransferase with respect to triacylglycerol synthesis in cultured rat hepatocytes was evaluated by following the inhibition of the overall synthetic flux by 2-bromooctanoate acting as an inhibitor of the diacylglycerol acyltransferase step. The flux-control coefficient
Chromatograms
suitable for GC자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.