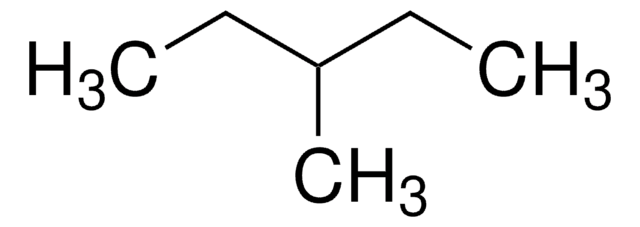
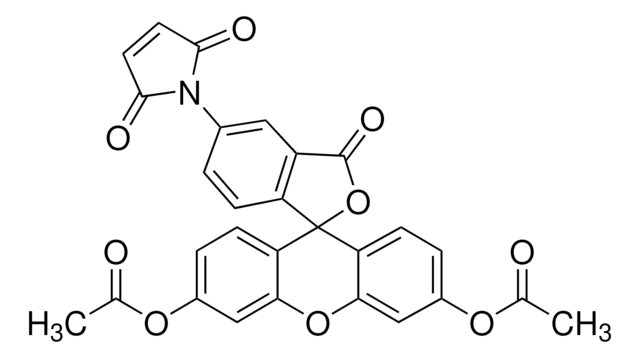
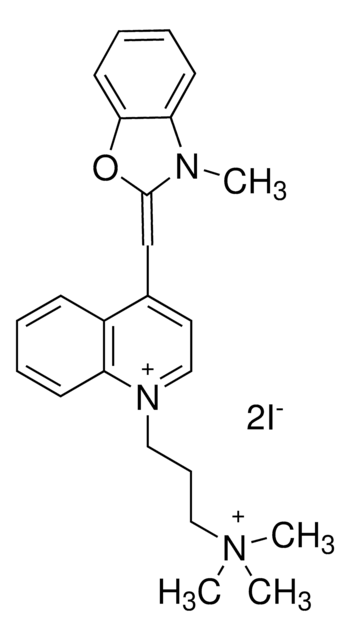
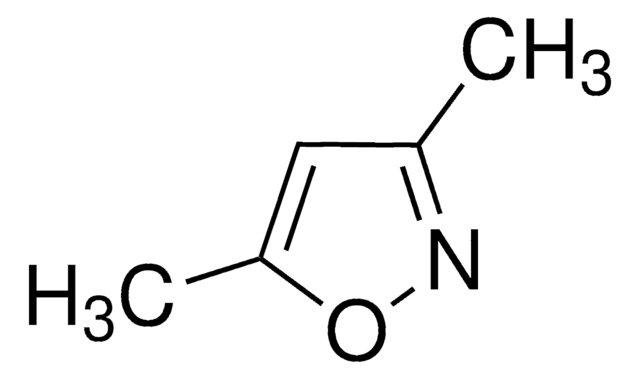
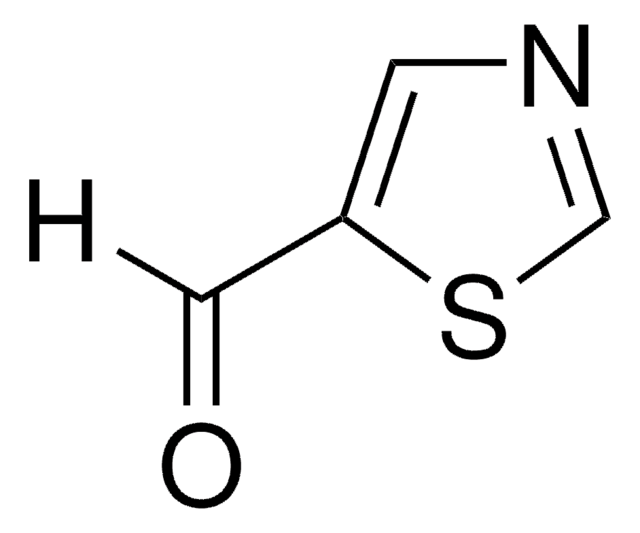
추천 제품
분석
98%
refractive index
n20/D 1.425 (lit.)
bp
69-70 °C (lit.)
mp
−87-−84 °C (lit.)
density
1.05 g/mL at 25 °C (lit.)
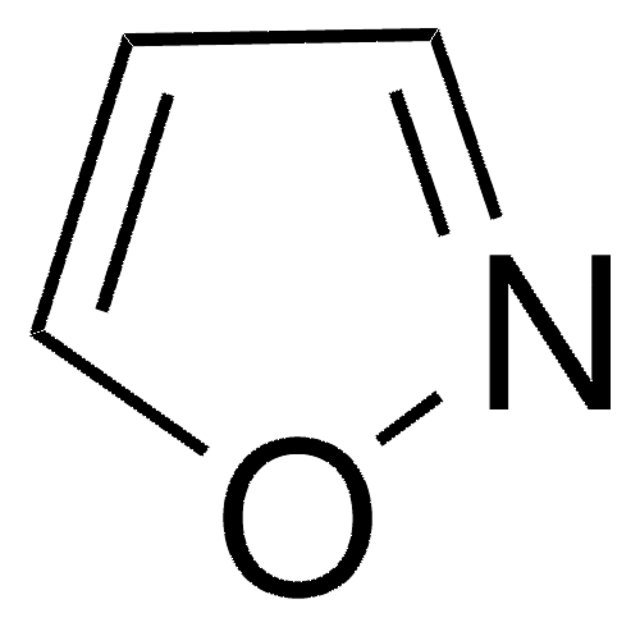
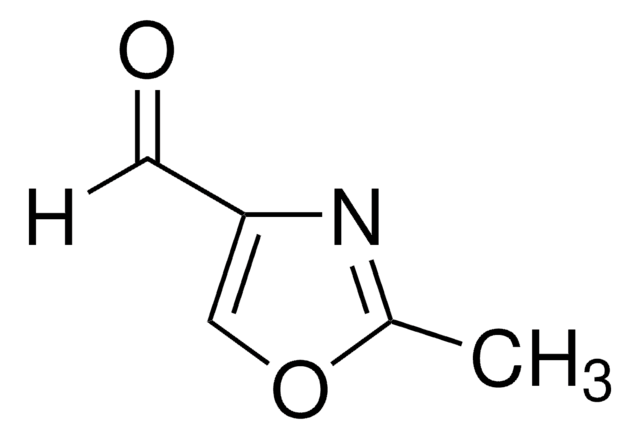
SMILES string
c1cocn1
InChI
1S/C3H3NO/c1-2-5-3-4-1/h1-3H
InChI key
ZCQWOFVYLHDMMC-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Oxazole is the parent molecule for a large class of heterocyclic aromatic compounds. It is a weak base that can be used as an electron-deficient diene in the Diels-Alder cycloaddition reaction. It undergoes nitration, sulfonation, halogenation, Friedel-Crafts alkylation, and acylation.
애플리케이션
Oxazole can be used:
- in the intramolecular Diels–Alder (IMDA) cycloaddition reaction to synthesis natural products
- as a precursor in the ring opening, nucleophilic addition and recyclization as well as [2 + 2], [3 + 2], and [4 + 2] cycloaddition reactions
신호어
Danger
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Eye Dam. 1 - Flam. Liq. 2
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point (°F)
66.2 °F - closed cup
Flash Point (°C)
19 °C - closed cup
개인 보호 장비
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
이미 열람한 고객
Maryna V Kachaeva et al.
Computational biology and chemistry, 74, 294-303 (2018-04-27)
Based on modern literature data about biological activity of E7010 derivatives, a series of new sulfonamides as potential anticancer drugs were rationally designed by QSAR modeling methods Сlassification learning QSAR models to predict the tubulin polymerization inhibition activity of novel
Haseen Ahmad et al.
European journal of medicinal chemistry, 208, 112759-112759 (2020-09-05)
Oxazole derivatives are important medicinal compounds which are inhibitors of various enzymes such as NPP1, NPP2, NPP3, tyrosine kinase, dipeptidyl-peptidase IV, cyclooxygenase-2, and protein tyrosine phosphatase. In this study, an extensive range of new biologically active biphenyl oxazole derivatives was
Dawid Siodłak et al.
The journal of physical chemistry. B, 118(9), 2340-2350 (2014-02-18)
Oxazole ring occurs in numerous natural peptides, but conformational properties of the amino acid residue containing the oxazole ring in place of the C-terminal amide bond are poorly recognized. A series of model compounds constituted by the oxazole-amino acids occurring
Ilya Lyagin et al.
Molecules (Basel, Switzerland), 24(13) (2019-06-30)
Mycotoxins are highly dangerous natural compounds produced by various fungi. Enzymatic transformation seems to be the most promising method for detoxification of mycotoxins. This review summarizes current information on enzymes of different classes to convert various mycotoxins. An in-depth analysis
Kristjan Bloudoff et al.
Proceedings of the National Academy of Sciences of the United States of America, 114(1), 95-100 (2016-12-21)
Nonribosomal peptide synthetases (NRPSs) are a family of multidomain, multimodule enzymes that synthesize structurally and functionally diverse peptides, many of which are of great therapeutic or commercial value. The central chemical step of peptide synthesis is amide bond formation, which
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.