추천 제품
분석
90%
반응 적합성
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: ligand
bp
111-113 °C/2.5 mmHg (lit.)
mp
48-56 °C (lit.)
작용기
phosphine
SMILES string
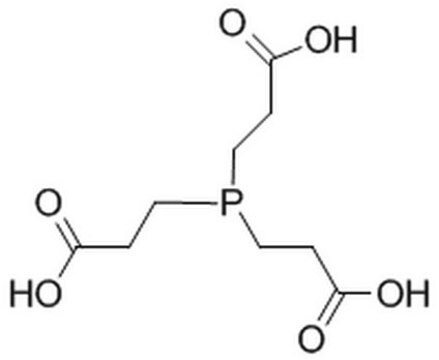
OCP(CO)CO
InChI
1S/C3H9O3P/c4-1-7(2-5)3-6/h4-6H,1-3H2
InChI key
JMXMXKRNIYCNRV-UHFFFAOYSA-N
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 3 Oral - Eye Dam. 1 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
이미 열람한 고객
Baobin Li et al.
Nature communications, 8(1), 666-666 (2017-09-25)
The O-linked β-N-acetyl glucosamine (O-GlcNAc) modification dynamically regulates the functions of numerous proteins. A single human enzyme O-linked β-N-acetyl glucosaminase (O-GlcNAcase or OGA) hydrolyzes this modification. To date, it remains largely unknown how OGA recognizes various substrates. Here we report
Christoph Krettler et al.
Analytical biochemistry, 474, 89-94 (2014-12-20)
Use of the reductant dithiothreitol (DTT) as a substrate for measuring vitamin K 2,3-epoxide reductase (VKOR) activity in vitro has been reported to be problematic because it enables side reactions involving the vitamin K1 2,3-epoxide (K1>O) substrate. Here we characterize
Rhushikesh A Kulkarni et al.
Nature chemical biology, 15(4), 391-400 (2019-02-06)
Hereditary cancer disorders often provide an important window into novel mechanisms supporting tumor growth. Understanding these mechanisms thus represents a vital goal. Toward this goal, here we report a chemoproteomic map of fumarate, a covalent oncometabolite whose accumulation marks the
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 177881-25G | 4061838753526 |
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.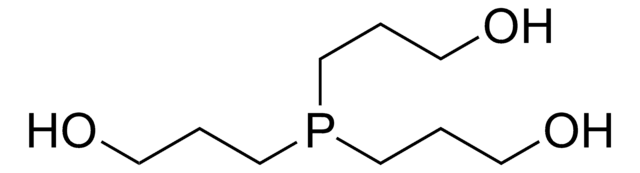
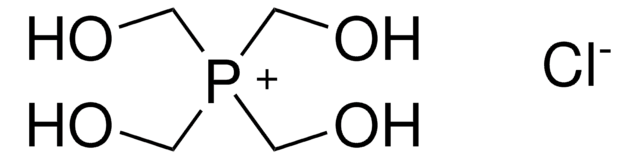


![Bis[tetrakis(hydroxymethyl)phosphonium] sulfate solution technical, 70-75% in H2O (T)](/deepweb/assets/sigmaaldrich/product/structures/236/818/d029a904-00ee-46ad-9fe2-2369f9894cc4/640/d029a904-00ee-46ad-9fe2-2369f9894cc4.png)
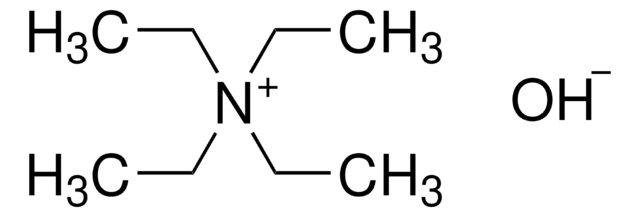


![Tetrakis[tris(dimethylamino)phosphoranylidenamino]phosphonium chloride 95%](/deepweb/assets/sigmaaldrich/product/structures/160/963/9dd6d457-17b2-44dc-8ea2-d3c0475b3664/640/9dd6d457-17b2-44dc-8ea2-d3c0475b3664.png)