추천 제품
Quality Level
분석
95%
refractive index
n20/D 1.406 (lit.)
bp
85 °C (lit.)
density
0.87 g/mL at 25 °C (lit.)
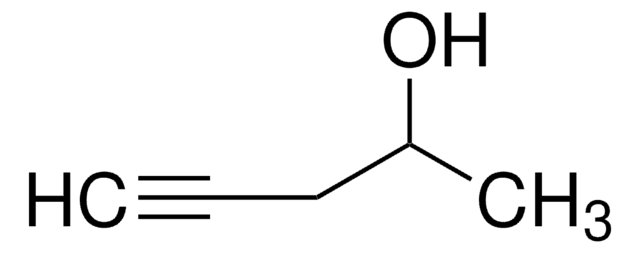
작용기
ketone
SMILES string
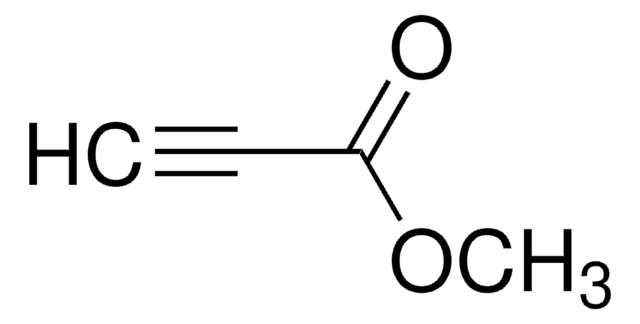
CC(=O)C#C
InChI
1S/C4H4O/c1-3-4(2)5/h1H,2H3
InChI key
XRGPFNGLRSIPSA-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
3-Butyn-2-one undergoes asymmetric double-Michael reaction with ortho-tosylamidophenyl malonate catalyzed by chiral aminophosphines to yield indolines. It undergoes double Michael reaction with nitrogen-containing tethered diacid to give pipecolic acid derivatives.
애플리케이션
3-Butyn-2-one was used in the synthesis of clerodane diterpenoid (+/-)-sacacarin. It was used as substrate in stereoselective, conjugate arylation mediated by gallium(III) chloride leading to (E)-α,β-unsaturated ketones.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 2 Oral - Eye Irrit. 2 - Flam. Liq. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point (°F)
30.2 °F - closed cup
Flash Point (°C)
-1 °C - closed cup
개인 보호 장비
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
이미 열람한 고객
Synlett, 809-809 (2007)
R B Grossman et al.
Organic letters, 3(25), 4027-4030 (2001-12-12)
[reaction: see text] The putative structure of the naturally occurring clerodane diterpenoid (+/-)-sacacarin has been prepared in only 10 steps, six of which are C-C bond-forming steps, in a chemo-, regio-, and diastereoselective manner. The key part of the synthesis
F Hughes et al.
Organic letters, 3(18), 2911-2914 (2001-09-01)
[reaction: see text]. Nitrogen-containing tethered diacids, easily prepared by reductive alkylation of diethyl aminomalonate or ethyl cyanoglycinate, undergo double Michael reactions with 3-butyn-2-one to give highly functionalized and substituted piperidines (pipecolic acid derivatives) with surprisingly high stereoselectivity. The heterocyclic double
San N Khong et al.
Molecules (Basel, Switzerland), 17(5), 5626-5650 (2012-05-15)
The bisphosphine-catalyzed double-Michael addition of dinucleophiles to electron-deficient acetylenes is an efficient process for the synthesis of many nitrogen-containing heterocycles. Because the resulting heterocycles contain at least one stereogenic center, this double-Michael reaction would be even more useful if an
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.