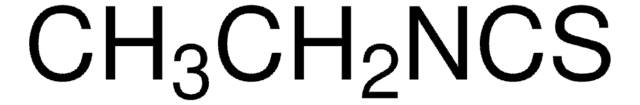추천 제품
Quality Level
분석
≥95.0% (GC)
형태
solid
bp
230-235 °C (lit.)
mp
39-41 °C (lit.)
39-41 °C
solubility
diethyl ether: soluble 0.5 g/10 mL, clear to very faintly turbid, colorless to almost colorless
작용기
phenyl
thiocyanate
thioether
저장 온도
2-8°C
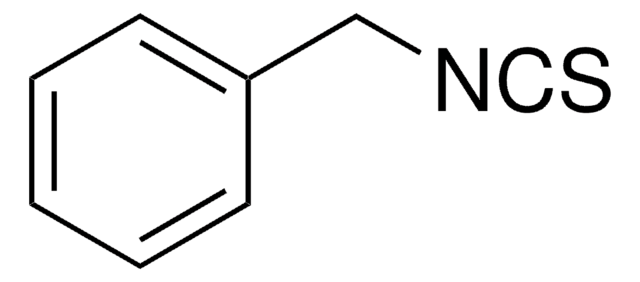
SMILES string
N#CSCc1ccccc1
InChI
1S/C8H7NS/c9-7-10-6-8-4-2-1-3-5-8/h1-5H,6H2
InChI key
ABNDFSOIUFLJAH-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Benzyl thiocyanate undergoes regioselective bond dissociation during its electrochemical reduction in acetonitrile at an inert electrode. It is added to stimulate the chlortetracycline biosynthesis during industrial fermentations. It undergoes biotransformation into dibenzyl disulphide by Streptomyces aureofaciens.
애플리케이션
Benzyl thiocyanate was used to study the effects of various dietary compounds on the α-hydroxylation of N-nitrosopyrrolidine in male F344 rats in vitro.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Aquatic Chronic 3
보충제 위험성
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point (°F)
230.0 °F
Flash Point (°C)
110 °C
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
S Sugie et al.
Carcinogenesis, 15(8), 1555-1560 (1994-08-01)
The effects of two aromatic thiocyanates, benzyl thiocyanate (BTC) and benzyl isothiocyanate (BITC), on methylazoxymethanol (MAM) acetate-induced intestinal carcinogenesis were examined using female ACI/N rats. Starting at 5 weeks of age, animals were fed diets containing 100 or 400 p.p.m.
F L Chung et al.
Cancer research, 44(7), 2924-2928 (1984-07-01)
Male F344 rats were pretreated with various dietary compounds, and the effects of pretreatment on the in vitro alpha-hydroxylation of N-nitrosopyrrolidine or N'-nitrosonornicotine were determined in assays with liver microsomes or cultured esophagus, respectively. Dietary compounds included phenols, cinnamic acids
Abdelaziz Houmam et al.
Journal of the American Chemical Society, 125(42), 12676-12677 (2003-10-16)
The electrochemical reduction of benzyl thiocyanate and p-nitrobenzyl thiocyanate was investigated in acetonitrile at an inert electrode. These two compounds reveal a change in the reductive cleavage mechanism, and more interestingly, they show a clear-cut example of a regioselective bond
J Fuska et al.
Letters in applied microbiology, 19(3), 124-125 (1994-09-01)
Benzyl thiocyanate, a specific stimulator of chlortetracycline biosynthesis, was transformed into dibenzyl disulphide by Streptomyces aureofaciens. The disulphide stimulated chlortetracycline production to a lesser extent than did benzyl thiocyanate.
X M Li et al.
Applied microbiology and biotechnology, 57(5-6), 717-724 (2002-01-10)
Changes in synthesis and abundance of proteins associated with chlortetracycline (CTC) production in Streptomyces aureofaciens were investigated by two-dimensional polyacrylamide gel electrophoresis of proteins pulse-labelled in vivo with L-[35S]methionine. Eleven individual protein spots were selected as being related to formation
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.