추천 제품
분석
≥95.0%
refractive index
n20/D 1.561 (lit.)
n20/D 1.563
bp
90-92 °C/15 mmHg (lit.)
density
1.174 g/mL at 25 °C (lit.)
저장 온도
2-8°C
SMILES string
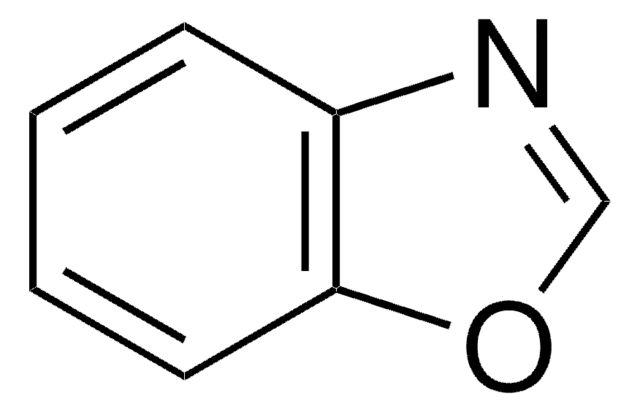
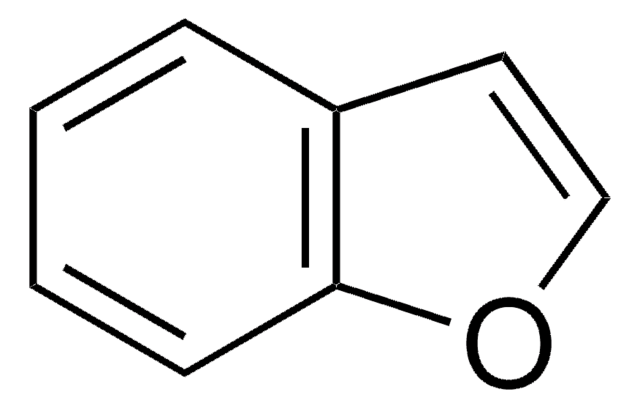
c1ccc2oncc2c1
InChI
1S/C7H5NO/c1-2-4-7-6(3-1)5-8-9-7/h1-5H
InChI key
KTZQTRPPVKQPFO-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
관련 카테고리
일반 설명
1,2-Benzisoxazole derivative zonisamide is a novel antiepileptic drug and is effective for the treatment of partial seizures. 1,2-Benzisoxazole is a potential substrates of rabbit liver aldehyde oxidase.
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point (°F)
186.8 °F - closed cup
Flash Point (°C)
86 °C - closed cup
개인 보호 장비
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
D H Peters et al.
Drugs, 45(5), 760-787 (1993-05-01)
Zonisamide is a 1,2 benzisoxazole derivative and the first agent of this chemical class to be developed as an antiepileptic drug. It has shown activity in various animal models of epilepsy, and although a detailed mode of action awaits clarification
W Gristwood et al.
Xenobiotica; the fate of foreign compounds in biological systems, 18(8), 949-954 (1988-08-01)
1. Twelve oxygen and sulphur azaheterocycles were studied as potential substrates of rabbit liver aldehyde oxidase. Only benzoxazole and 1,2-benzisoxazole were found to be substrates. 2. Nine of the compounds inhibited the oxidation of quinazoline by aldehyde oxidase and in
Nan-Sook Hong et al.
Nature communications, 9(1), 3900-3900 (2018-09-27)
Developments in computational chemistry, bioinformatics, and laboratory evolution have facilitated the de novo design and catalytic optimization of enzymes. Besides creating useful catalysts, the generation and iterative improvement of designed enzymes can provide valuable insight into the interplay between the
Avneet Kaur et al.
Archiv der Pharmazie, 351(6), e1800008-e1800008 (2018-05-10)
A series of N-(2-(3,5-dimethoxyphenyl)benzoxazole-5-yl)benzamide derivatives (3am) was synthesized and evaluated for their in vitro inhibitory activity against COX-1 and COX-2. The compounds with considerable in vitro activity (IC50 < 1 μM) were evaluated in vivo for their anti-inflammatory potential by the carrageenan-induced
Min Bao et al.
International journal of biological macromolecules, 137, 537-544 (2019-06-25)
Studies on endo-inulinases from yeast are scarce, compared to those from other microbial sources. In this study, a novel endo-inulinase from Lipomyces starkeyi NRRL Y-11557 was identified, expressed in its soluble form, and characterized its physicochemically properties, together with its
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.