SBR00031
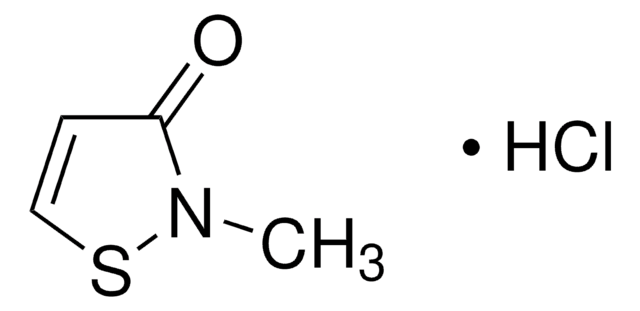
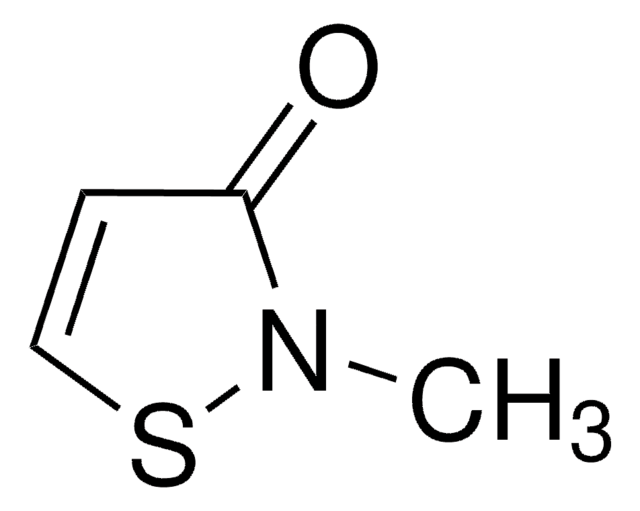
MIT HCl Ready Made Solution
(10 mg MIT/mL)
Synonym(s):
2-Methyl-1,2-thiazol-3-one, Methylisothiazolinone HCl, Methylisothiazolinone hydrochloride
About This Item
Recommended Products
form
liquid
Quality Level
concentration
(10 mg MIT/mL)
1%
antibiotic activity spectrum
Gram-negative bacteria
Gram-positive bacteria
Mode of action
protein synthesis | interferes
storage temp.
2-8°C
InChI
1S/C4H5NOS.ClH/c1-5-4(6)2-3-7-5;/h2-3H,1H3;1H
InChI key
SJXPQSRCFCPWQQ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- in research on the effects of the N-methyl D-aspartate (NMDA) receptor antagonist kynurenic acid on human cortical development
- as a cytotoxic substance to investigate its effect on bronchial epithelial cells (BEAS-2B cells) and its role in apoptotic cell death
- to research the effects of tyrosine phosphorylation on focal adhesion kinase (FAK) activity in the development of neural axons and dendrites
Biochem/physiol Actions
Activity Spectrum: Active against Gram-positive and Gram-negative bacteria, as well as fungi, when used in combination with methylchloroisothiazolinone (MCI)
Features and Benefits
- Ready available solution reduce the need for preparation time
- Commonly used in Cell Biology and Biochemical applications
- High quality antibiotic suitable for mulitple research applications
Other Notes
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Chronic 3 - Eye Irrit. 2 - Skin Irrit. 2 - Skin Sens. 1
Storage Class Code
12 - Non Combustible Liquids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
JAN Code
SBR00031-VAR:
SBR00031-10ML:4548173372068
SBR00031-BULK:
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service