ML0015
Aspartate Metabolite Library
Synonym(s):
Aspartic acid Metabolite Library
About This Item
Recommended Products
General description
In the L-conformation, aspartic acid is a building block in the production of proteins, as well as aiding in many bodily functions, including the urea cycle, gluconeogenesis, and Krebs Cycle, a process that generates adenosine triphosphate (ATP). Aspartic acid also works as a neurotransmitter. The D-Aspartate conformation is linked to neurogenesis and endocrine systems.
Application
Biochem/physiol Actions
- Aspartate is synthesized by transamination of oxaloacetate through the actions of Aspartate aminotransferase and pyridoxal 5′- phosphate. Aspartyl-tRNA synthase can then couple the aspartate to aspartyl tRNA for protein synthesis.
- Aspartate carries the reducing equivalents in the mitochondrial Malate-Aspartate shuttle, which uses the ready interconversion of aspartate and oxaloacetate.
- N-acetylaspartate synthase, present in the cytoplasm, converts aspartate to N-acetylaspartate, a brain metabolite that regulates dopamine.
- Asparagine is biosynthesized by Asparagine synthetase from aspartate, glutamine, and ATP. Asparagine is involved in the metabolic control of cell functions in nerve and brain tissue.
- Arginosuccinic acid is synthesized from aspartate, citrulline and ATP through the action of Argininosuccinate synthase, one of the enzymes of the urea cycle. In this metabolic pathway, neurotoxic ammonia, produced by protein catabolism, is converted into urea in the liver.
- Fumaric acid is synthesized from Argininosuccinic acid via an Argininosuccinate lyase, which is an enzyme in the Citric Acid Cycle.
- Inosinic acid, aspartic acid and GTP are interconverted to GDP and AMP by the Adenylosuccinate synthetase isozyme 1. This process is involved in the purine nucleotide cycle which regulates nucleotides levels in various tissues.
- Aspartate transcarbamoylase catalyzes the synthesis of N-carbamoyl-L-aspartate from carbamoyl phosphate and aspartate that are involved in the de novo biosynthesis of pyrimidines.
- Beta alanine is formed by decarboxylation of aspartate by Glutamate decarboxylase 1 in the cytoplasm.
- L-aspartate is converted to D-aspartate through the action of a D-aspartate racemase. D-aspartate contributes to the synthesis and release of glucocorticoids, prolactin, oxytocin, and steroids. D-aspartate plays an important role in the brain activity of mammals.
Components
Kit Components Also Available Separately
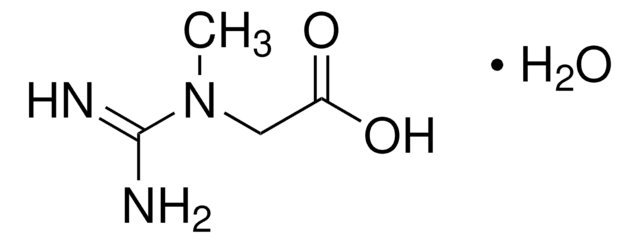
- A2252Adenosine 5′-monophosphate monohydrate, from yeast, ≥97%SDS
- A2383Adenosine 5′-triphosphate disodium salt hydrate, Grade I, ≥99%, from microbialSDS
- A5707Argininosuccinic acid disodium salt hydrate, ≥80%SDS
- 146064β-Alanine, 99%SDS
- C4135Carbamyl phosphate disodium salt, ≥80%SDS
- C7629L-Citrulline, ≥98% (TLC)SDS
- 219096D-Aspartic acid, ReagentPlus®, 99%SDS
- F6625Flavin adenine dinucleotide disodium salt hydrate, ≥95% (HPLC), powderSDS
- 47910Fumaric acid, ≥99.0% (T)SDS
- G7252Guanosine 5′-diphosphate tris salt from Saccharomyces cerevisiae, Type VI, ≥92.5%SDS
- G9002Guanosine 5′-triphosphate tris salt, ≥93% (HPLC), powderSDS
- I2879Inosine 5′-monophosphate from Saccharomyces cerevisiae, ≥98%SDS
- A5006L-Arginine, reagent grade, ≥98%SDS
- A0884L-Asparagine, ≥98% (HPLC)SDS
- 11189L-Aspartic acid, BioUltra, ≥99.5% (T)SDS
- G1251L-Glutamic acid, ReagentPlus®, ≥99% (HPLC)SDS
- G3126L-Glutamine, ReagentPlus®, ≥99% (HPLC)SDS
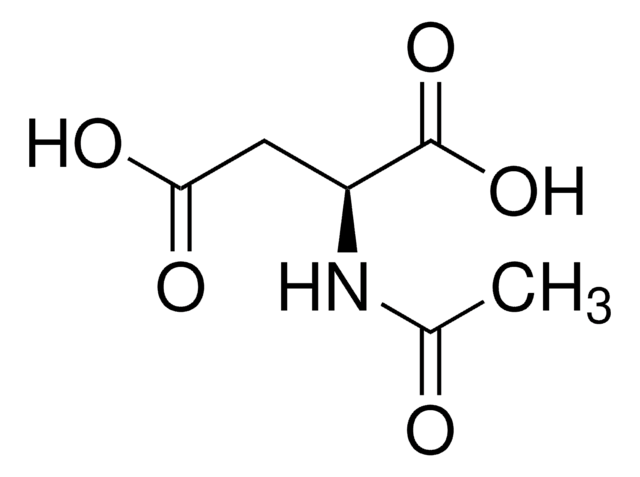
- 00920N-Acetyl-L-aspartic acid, ≥99.0% (T)SDS
- O4126Oxaloacetic acid, ≥97% (HPLC)SDS
- K1750α-Ketoglutaric acid, ≥98.5% (NaOH, titration)SDS
- P9255Pyridoxal 5′-phosphate hydrate, ≥98%SDS
- P8010Sodium pyrophosphate tetrabasic, ≥95%SDS
- 69037Ureidosuccinic acid, 98.0-102.0% (T)SDS
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1
Storage Class Code
11 - Combustible Solids
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
PDSCL
Please refer to KIT Component information
PRTR
Please refer to KIT Component information
FSL
Please refer to KIT Component information
ISHL Indicated Name
Please refer to KIT Component information
ISHL Notified Names
Please refer to KIT Component information
Cartagena Act
Please refer to KIT Component information
JAN Code
キットコンポーネントの情報を参照してください
Choose from one of the most recent versions:
Certificates of Analysis (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documents section.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service