656631
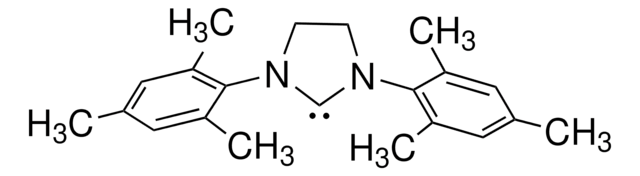
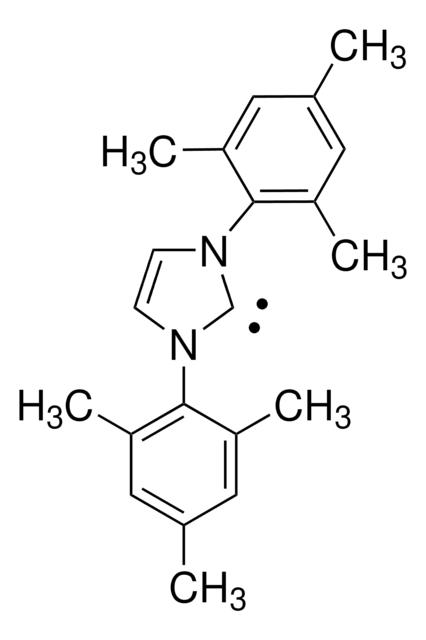
1,3-Bis(2,4,6-trimethylphenyl)imidazolinium chloride
95%
Synonym(s):
1,3-Dimesitylimidazolidinium chloride, 4,5-Dihydro-1,3-bis(2,4,6-trimethylphenyl)-1H-imidazolium chloride, 4,5-Dihydro-1,3-dimesityl-1H-imidazolium chloride, N,N′-(2,4,6-Trimethylphenyl)dihydroimidazolium chloride
About This Item
Recommended Products
Quality Level
Assay
95%
reaction suitability
reagent type: ligand
mp
280-286 °C
SMILES string
[Cl-].Cc1cc(C)c(N2CC[N+](=C2)c3c(C)cc(C)cc3C)c(C)c1
InChI
1S/C21H27N2.ClH/c1-14-9-16(3)20(17(4)10-14)22-7-8-23(13-22)21-18(5)11-15(2)12-19(21)6;/h9-13H,7-8H2,1-6H3;1H/q+1;/p-1
InChI key
COGMCBFILULEOS-UHFFFAOYSA-M
Related Categories
General description
Application
Precursor to an N-heterocyclic carbene catalysts used for:
- A regioselective cycloadditon of terminal acetylenes with azides leading to 1,4-disubstitutedtriazoles. Internal acetylenes can also be used with this catalyst.
- Markovnikov-type hydration of terminal alkynes
- Hydrosilylation of ketones and cycloaddition of azides and alkynes
- Suzuki-Miyaura reactions
- Luminescence experiments
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
JAN Code
656631-BULK:
656631-1G:
656631-VAR:
656631-5G:
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Rapid progress in cross-coupling reactions of unactivated substrates catalyzed by metal complexes has transformed the chemical market-place through introduction of an extensive library of achiral and chiral phosphine ligands.
A wide range of NHC ligands are commonly available which exhibit high activities.
Emerging class of privileged ligands
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service


![Chloro[1,3-Bis(2,4,6-trimethylphenyl)imidazol-2-ylidene]copper(I) 95%](/deepweb/assets/sigmaaldrich/product/structures/160/888/97509eeb-0719-4853-aaae-8a9d02f4f7ad/640/97509eeb-0719-4853-aaae-8a9d02f4f7ad.png)
![Chloro[1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene]copper(I)](/deepweb/assets/sigmaaldrich/product/structures/199/763/44637b2e-b87c-42a3-abc3-3985b6cd7d5d/640/44637b2e-b87c-42a3-abc3-3985b6cd7d5d.png)