L8543
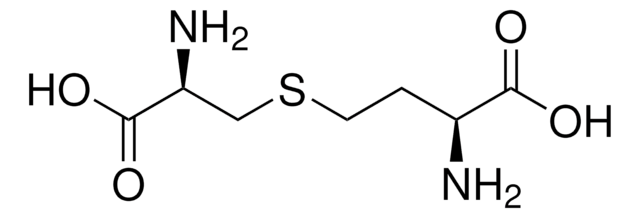
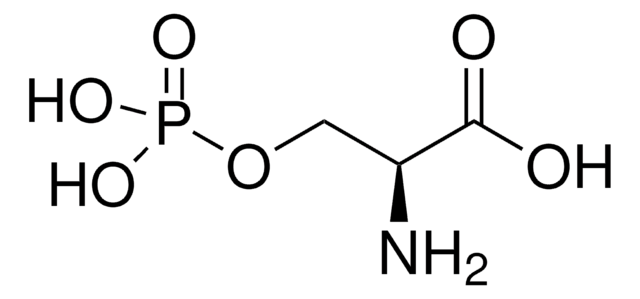
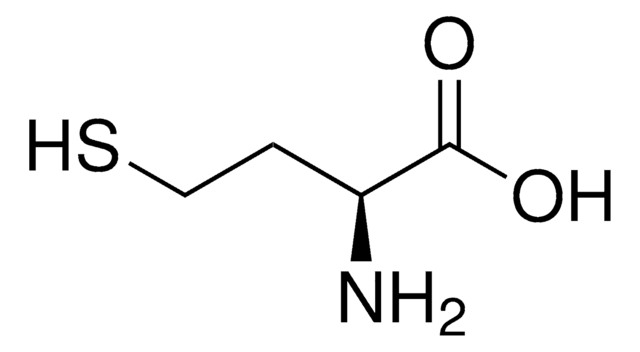
DL-Lanthionine
≥98% (TLC)
Sinonimo/i:
DL-3,3′-thiodi-Alanine, S-(2-amino-2-carboxyethyl)-DL-Cysteine
About This Item
Prodotti consigliati
product name
DL-Lanthionine, ≥98% (TLC)
Livello qualitativo
Saggio
≥98% (TLC)
Forma fisica
powder
Colore
white
Solubilità
1 M HCl: soluble
applicazioni
detection
InChI
1S/C6H12N2O4S/c7-3(5(9)10)1-13-2-4(8)6(11)12/h3-4H,1-2,7-8H2,(H,9,10)(H,11,12)
DWPCPZJAHOETAG-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Applicazioni
- Synthesis of the lantibiotic lactocin S using peptide cyclizations on solid phase.: This research highlights innovative methods for synthesizing peptide-based antibiotics like lactocin S, utilizing dl-lanthionine to form crucial thioether crosslinks that enhance the activity and stability of these peptides (Ross et al., 2010).
- Cystathionine gamma-lyase of Streptomyces phaeochromogenes.: This research documents the isolation and characterization of an enzyme that processes cystathionine, which involves dl-lanthionine as a structural analog, revealing its role in bacterial physiology and potential applications in biotechnology (Nagasawa et al., 1984).
- The availability of dl-lanthionine for the promotion of growth in young rats when added to a cystine- and methionine-deficient diet.: This historical study explores the nutritional value of dl-lanthionine, investigating its ability to substitute for essential amino acids in growth diets, which helps in understanding its potential use in nutritional supplements (Jones et al., 1948).
Azioni biochim/fisiol
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.