L5768
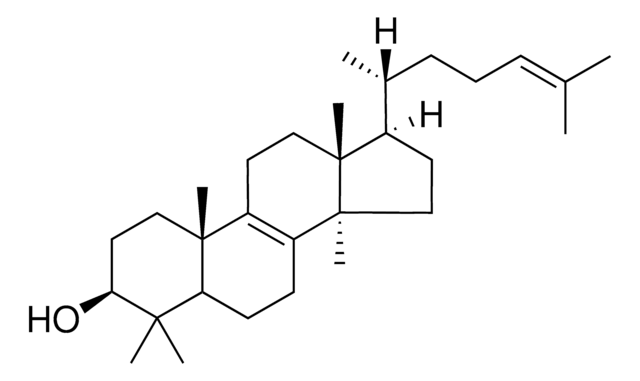
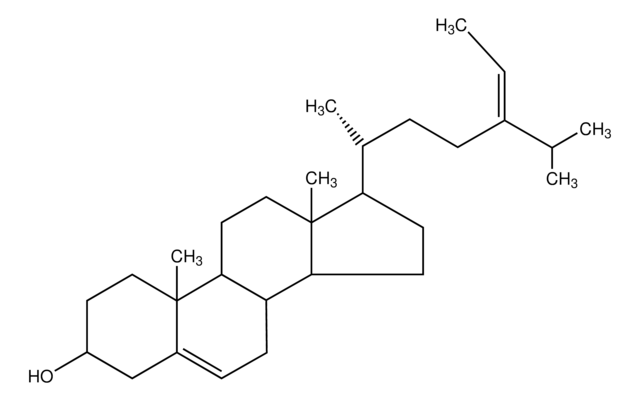
Lanosterol
≥93%, powder
Sinonimo/i:
3β-Hydroxy-8,24-lanostadiene, 8,24-Lanostadien-3β-ol
About This Item
Prodotti consigliati
Saggio
≥93%
Stato
powder
Colore
white to off-white
Temperatura di conservazione
−20°C
Stringa SMILE
[H][C@@]1(CC[C@@]2(C)C3=C(CC[C@]12C)[C@@]4(C)CC[C@H](O)C(C)(C)[C@]4([H])CC3)[C@H](C)CC\C=C(/C)C
InChI
1S/C30H50O/c1-20(2)10-9-11-21(3)22-14-18-30(8)24-12-13-25-27(4,5)26(31)16-17-28(25,6)23(24)15-19-29(22,30)7/h10,21-22,25-26,31H,9,11-19H2,1-8H3/t21-,22-,25+,26+,28-,29-,30+/m1/s1
CAHGCLMLTWQZNJ-BQNIITSRSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Descrizione generale
Applicazioni
- as a standard in HPLC for the quantification in testis samples
- in S-adenosyl-L-methionine:Δ24-sterol-C-methyltransferase (SMT) assay
- to treat wild-type cells growing in rich medium to know its effects on Sre1 protein
Azioni biochim/fisiol
Lanosterol serves as an endogenous selective modulator of macrophage immunity.
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 1
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Articoli
Biosynthesis of cholesterol generally takes place in the endoplasmic reticulum of hepatic cells and begins with acetyl- CoA, which is mainly derived from an oxidation reaction in the mitochondria. Acetyl-CoA and acetoacetyl-CoA are converted to 3-hydroxy- 3-methylglutaryl-CoA (HMG-CoA) by HMG-CoA synthase.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.