A6011
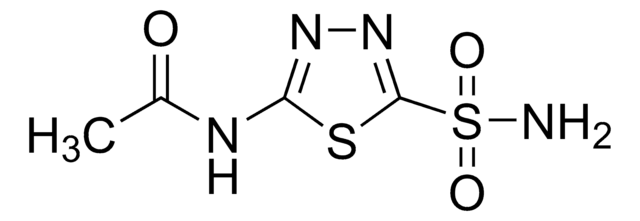
Acetazolamide
≥99%, powder
Sinonimo/i:
5-Acetamido-1,3,4-thiadiazole-2-sulfonamide, N-(5-Sulfamoyl-1,3,4-thiadiazol-2-yl)acetamide, N-(5-[Aminosulfonyl]-1,3,4-thiadiazol-2-yl)acetamide
About This Item
Prodotti consigliati
Origine biologica
synthetic
Saggio
≥99%
Stato
powder
pKa
7.2
Punto di fusione
258-259 °C
Solubilità
1 M NH4OH: 50 mg/mL
DMSO: soluble
methanol and ethanol: slightly soluble
Stringa SMILE
CC(=O)Nc1nnc(s1)S(N)(=O)=O
InChI
1S/C4H6N4O3S2/c1-2(9)6-3-7-8-4(12-3)13(5,10)11/h1H3,(H2,5,10,11)(H,6,7,9)
BZKPWHYZMXOIDC-UHFFFAOYSA-N
Informazioni sul gene
human ... CA1(759) , CA12(771) , CA14(23632) , CA2(760) , CA3(761) , CA4(762) , CA5A(763) , CA5B(11238) , CA9(768)
mouse ... Car13(71934) , Car5a(12352)
rat ... Car2(54231) , Car4(29242)
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Applicazioni
- to study its protective effect on steatotic liver grafts against cold ischemia reperfusion injury{49)
- to determine its ability to bind isolated porcine retinal pigment epithelium (RPE) melanin by cassette dosing and rapid equilibrium dialysis inserts
- to validate the in vitro gastrulation model of P19C5 stem cells for developmental toxicity screening assays
- to study its inhibitory effect on melanogenesis through enzyme kinetic, in vitro, in vivo and in silico analyses in zebrafish and in A375 human melanoma cells
Azioni biochim/fisiol
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Skin Irrit. 2
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.