PHR1389
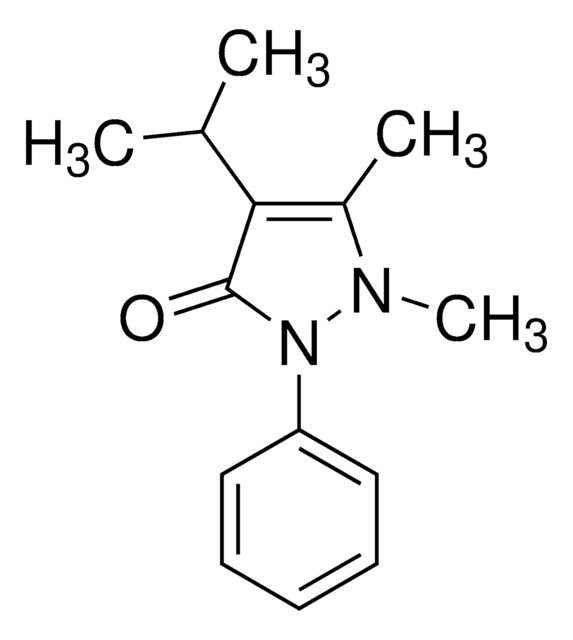
Propyphenazone
Pharmaceutical Secondary Standard; Certified Reference Material
Sinonimo/i:
4-Isopropylantipyrine, 1,2-Dihydro-1,5-dimethyl-4-(1-methylethyl)-2-phenyl-3H-pyrazol-3-one, 1,2-Dihydro-4-isopropyl-1,5-dimethyl-2-phenyl-3H-pyrazol-3-one, Isopropylphenazone, Propyphenazone
About This Item
Prodotti consigliati
Grado
certified reference material
pharmaceutical secondary standard
Livello qualitativo
agenzia
traceable to Ph. Eur. P3750000
Famiglia di API
propyphenazone
CdA
current certificate can be downloaded
tecniche
HPLC: suitable
gas chromatography (GC): suitable
applicazioni
pharmaceutical (small molecule)
Formato
neat
Temperatura di conservazione
2-8°C
Stringa SMILE
O=C1C(C(C)C)=C(C)N(C)N1C2=CC=CC=C2
InChI
1S/C14H18N2O/c1-10(2)13-11(3)15(4)16(14(13)17)12-8-6-5-7-9-12/h5-10H,1-4H3
PXWLVJLKJGVOKE-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Applicazioni
Risultati analitici
Altre note
Nota a piè di pagina
Prodotti correlati
Scegli una delle versioni più recenti:
Certificati d'analisi (COA)
Non trovi la versione di tuo interesse?
Se hai bisogno di una versione specifica, puoi cercare il certificato tramite il numero di lotto.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.