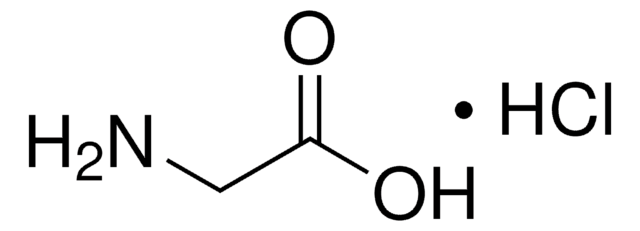
G6104
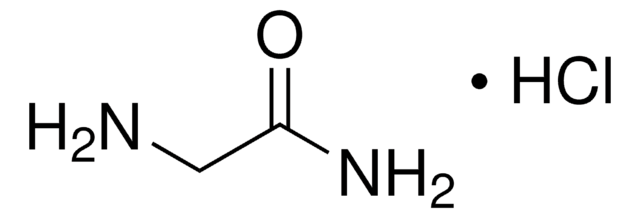
Glycinamide hydrochloride
98%
Sinonimo/i:
2-Aminoacetamide hydrochloride, Aminoacetamide hydrochloride, Glycine amide hydrochloride
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
NH2CH2CONH2 · HCl
Numero CAS:
Peso molecolare:
110.54
Beilstein:
3554199
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
98%
Punto di fusione
204 °C (dec.) (lit.)
Stringa SMILE
Cl.NCC(N)=O
InChI
1S/C2H6N2O.ClH/c3-1-2(4)5;/h1,3H2,(H2,4,5);1H
WKNMKGVLOWGGOU-UHFFFAOYSA-N
Applicazioni
Buffer useful in the physiological pH range.
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Gottfried K Schroeder et al.
Biochemistry, 46(13), 4037-4044 (2007-03-14)
As a model for mechanistic comparison with peptidyl transfer within the ribosome, the reaction of aqueous glycinamide with N-formylphenylalanine trifluoroethyl ester (fPhe-TFE) represents an improvement over earlier model reactions involving Tris. The acidity of trifluoroethanol (pKa 12.4) resembles that of
Eric Loeser et al.
Analytical chemistry, 79(14), 5382-5391 (2007-05-29)
When mobile-phase salt content is increased, cationic analytes often show increased retention. This effect is generally attributed to chaotropic or ion pairing effects. However, a cation exclusion mechanism could explain the same effects. In this study, experimental conditions were manipulated
Brett C Bookser et al.
Journal of medicinal chemistry, 48(24), 7808-7820 (2005-11-24)
4-(Phenylamino)-5-phenyl-7-(5-deoxy-beta-D-ribofuranosyl)pyrrolo[2,3-d]pyrimidine 1 and related compounds known as "diaryltubercidin" analogues are potent inhibitors of adenosine kinase (AK) and are orally active in animal models of pain such as the rat formalin paw model (GP3269 ED50= 6.4 mg/kg). However, the utility of
Xiaodong Jia et al.
The Journal of organic chemistry, 78(18), 9450-9456 (2013-08-21)
A catalytic α-sp(3) C-H oxidation of peptides and glycine amides was achieved under radical cation salt catalysis in the presence of O2, producing a series of substituted quinolines. The scope of this reaction shows good functional group tolerance and high
Hong Zhao et al.
Bioconjugate chemistry, 17(2), 341-351 (2006-03-16)
The utility of PEGylation for improving therapeutic protein pharmacology would be substantially expanded if the authentic protein drugs could be regenerated in vivo. Diminution of kinetic constants of both enzymes and protein ligands are commonly encountered following permanent bioconjugation with
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.