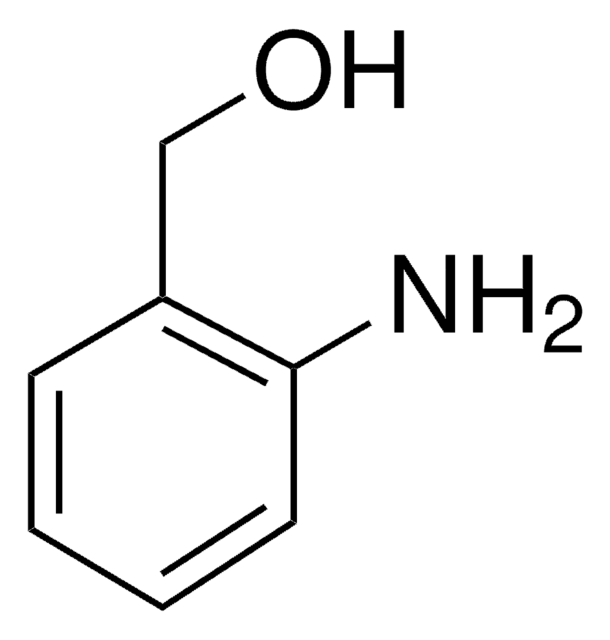
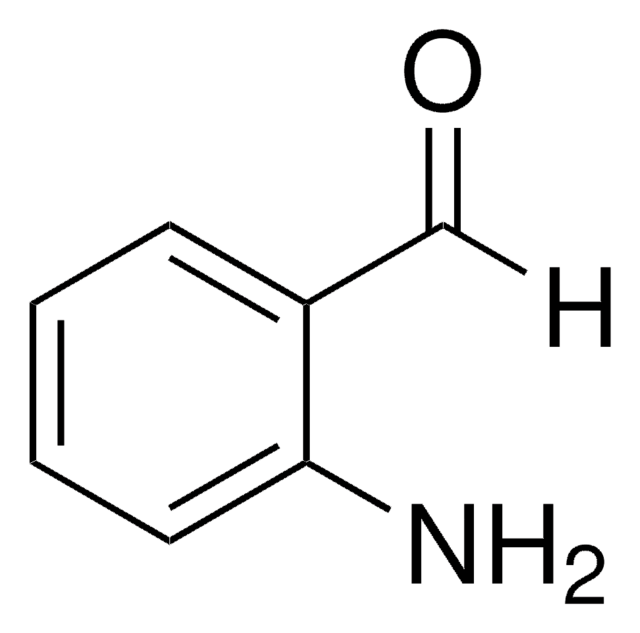
A38207
2-Aminoacetophenone hydrochloride
99%
Sinonimo/i:
ω-Aminoacetophenone hydrochloride, Phenacylamine hydrochloride
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
C6H5COCH2NH2 · HCl
Numero CAS:
Peso molecolare:
171.62
Beilstein:
3563173
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
99%
Stato
crystals
Punto di fusione
194 °C (dec.) (lit.)
Stringa SMILE
Cl.NCC(=O)c1ccccc1
InChI
1S/C8H9NO.ClH/c9-6-8(10)7-4-2-1-3-5-7;/h1-5H,6,9H2;1H
CVXGFPPAIUELDV-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
[Anorexigenic activity of various derivatives of alpha-aminoacetophenone].
M S Sánchez et al.
Archivos de farmacologia y toxicologia, 5(3), 165-168 (1979-12-01)
M J Bossard et al.
The Journal of biological chemistry, 265(10), 5640-5647 (1990-04-05)
A mechanism for beta-chlorophenethylamine inhibition of dopamine beta-monooxygenase has been postulated in which bound alpha-aminoacetophenone is generated followed by an intramolecular redox reaction to yield a ketone-derived radical cation as the inhibitory species (Mangold, J.B., and Klinman, J.P. (1984) J.
Frank Sporkert et al.
Forensic science international, 133(1-2), 39-46 (2003-05-14)
A sensitive and reproducible method for the quantitative determination of cathinone (CTN), norpseudoephedrine (NPE, cathine) and norephedrine (NE) from hair was developed. The compounds were extracted for 4 hours with phosphate buffer pH 2.0, followed by a standard solid phase
J B Mangold et al.
The Journal of biological chemistry, 259(12), 7772-7779 (1984-06-25)
Functionalization of the beta-carbon of phenethylamines has been shown to produce a new class of substrate/inhibitor of dopamine beta-monooxygenase. Whereas both beta-hydroxy- and beta- chlorophenethylamine are converted to alpha-aminoacetophenone at comparable rates, only the latter conversion is accompanied by concomitant
Yasumasa Iwai et al.
Chemical & pharmaceutical bulletin, 50(3), 441-443 (2002-03-26)
The new coupling reaction of phenacylamines with silylstannane and lithium diisopropylamide (LDA) is reported. The treatment of a phenacylamine iodide 1 with (trimethylsilyl)tributylstannane (Me3SiSnBu3) and cesium fluoride (CsF) gave a dimerization product 2 having no iodine atom. Reaction of 1
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.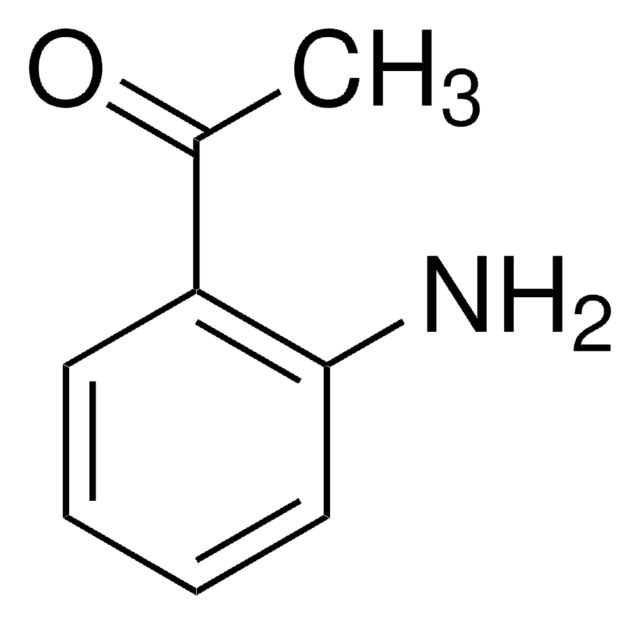
![2-[(1R)-1-Aminoethyl]-phenol AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/166/868/86b5a88f-7c6b-4634-aedc-c523a10e54aa/640/86b5a88f-7c6b-4634-aedc-c523a10e54aa.png)