779008
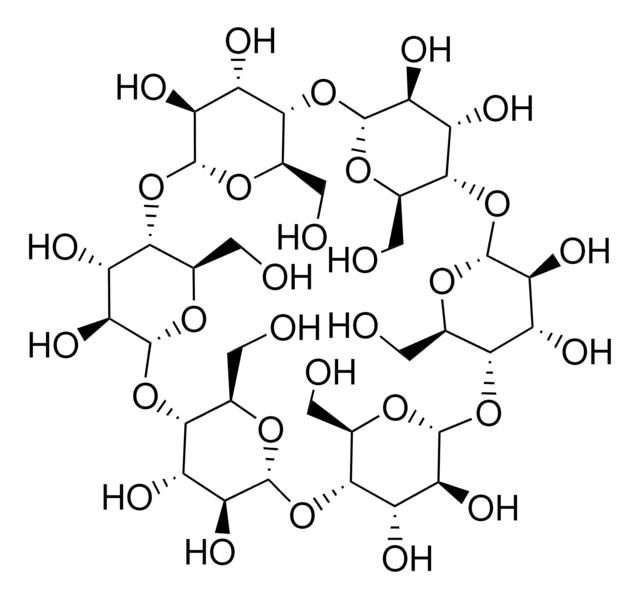
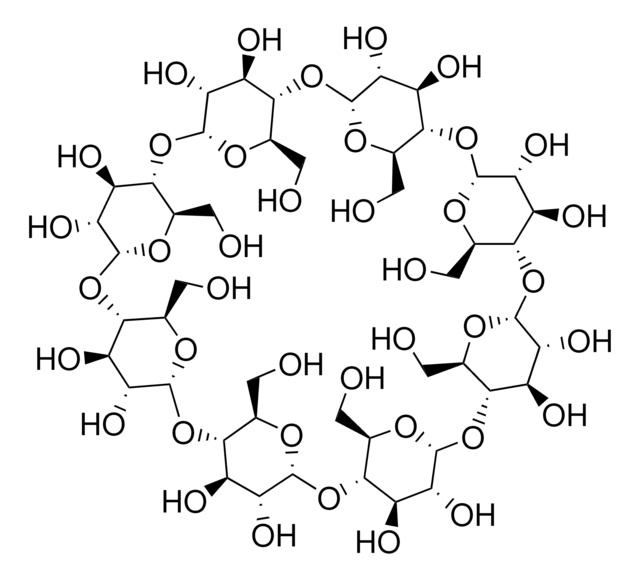
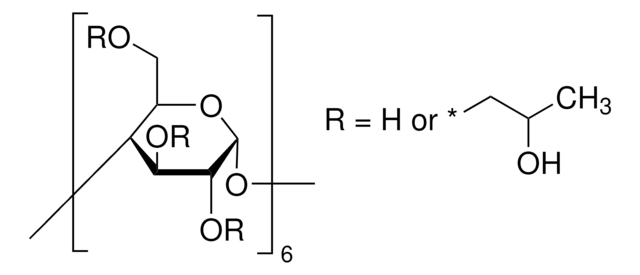
α-Cyclodextrin
Produced by Wacker Chemie AG, Burghausen, Germany, Life Science, 98.0-101.0% cyclodextrin basis (HPLC)
Sinonimo/i:
Cavamax® W6 Pharma, alpha-Cyclodextrin, α-Schardinger dextrin, Cyclohexaamylose, Cyclomaltohexaose
About This Item
Prodotti consigliati
Grado
Produced by Wacker Chemie AG, Burghausen, Germany, Life Science
Livello qualitativo
Saggio
98.0-101.0% cyclodextrin basis (HPLC)
Forma fisica
solid
Attività ottica
[α]/D 147.0 to 152.0° in H2O (USP)
Impurezze
≤0.20% reducing substances
≤0.25% β- and γ-cyclodextrin (each)
≤0.5% related substances
≤20 ppm residual solvents
≤5 ppm heavy metals (USP)
Residuo alla calcinazione
≤0.10% (USP)
Perdita
≤10.0% loss on drying
pH
5.0-8.0 (1% in solution)
Punto di fusione
>278 °C (dec.) (lit.)
Assorbimento
≤0.05 at 350-750 nm in solution at 1%
≤0.10 at 230-250 nm in solution at 1%
Compatibilità
positive for identity (Ph Eur)
Gruppo funzionale
ether
hydroxyl
Stringa SMILE
OC[C@H]1O[C@@H]2O[C@H]3[C@H](O)[C@@H](O)[C@H](O[C@@H]3CO)O[C@H]4[C@H](O)[C@@H](O)[C@H](O[C@@H]4CO)O[C@H]5[C@H](O)[C@@H](O)[C@H](O[C@@H]5CO)O[C@H]6[C@H](O)[C@@H](O)[C@H](O[C@@H]6CO)O[C@H]7[C@H](O)[C@@H](O)[C@H](O[C@@H]7CO)O[C@H]1[C@H](O)[C@H]2O
InChI
1S/C36H60O30/c37-1-7-25-13(43)19(49)31(55-7)62-26-8(2-38)57-33(21(51)15(26)45)64-28-10(4-40)59-35(23(53)17(28)47)66-30-12(6-42)60-36(24(54)18(30)48)65-29-11(5-41)58-34(22(52)16(29)46)63-27-9(3-39)56-32(61-25)20(50)14(27)44/h7-54H,1-6H2/t7-,8-,9-,10-,11-,12-,13-,14-,15-,16-,17-,18-,19-,20-,21-,22-,23-,24-,25-,26-,27-,28-,29-,30-,31-,32-,33-,34-,35-,36-/m1/s1
HFHDHCJBZVLPGP-RWMJIURBSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
- As a ligand in the complexation of nonionic surfactants and polyethylene glycols. α-cyclodextrin shows strong interaction with the surfactants that contains no benzene group.
- To protect linoleic acid from oxidation.
- In the synthesis of binuclear copper(II) complexes with cyclodextrins, which can be further used as a template for the preparation of copper nanoparticles incorporated on mesoporous silica.
Note legali
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 1
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.