676403
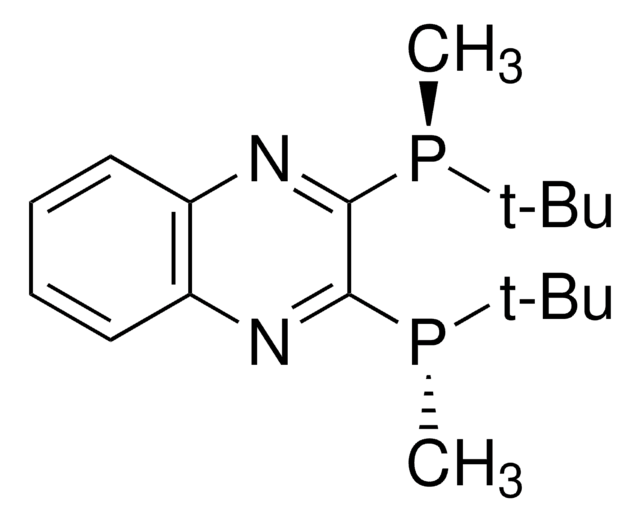
(R,R)-(–)-2,3-Bis(tert-butylmethylphosphino)quinoxaline
≥95%
Sinonimo/i:
(R) QuinoxP®
About This Item
Prodotti consigliati
Saggio
≥95%
Stato
solid
Punto di fusione
100-104 °C
Gruppo funzionale
phosphine
Stringa SMILE
CP(c1nc2ccccc2nc1P(C)C(C)(C)C)C(C)(C)C
InChI
1S/C18H28N2P2/c1-17(2,3)21(7)15-16(22(8)18(4,5)6)20-14-12-10-9-11-13(14)19-15/h9-12H,1-8H3/t21-,22-/m0/s1
DRZBLHZZDMCPGX-VXKWHMMOSA-N
Categorie correlate
Descrizione generale
Applicazioni
Caratteristiche e vantaggi
- It is not oxidized nor epimerized at ambient conditions in air
- Enantioselectivities are outstanding for various reaction paradigms
- Hydrogenations proceed under mild reaction conditions
- Low catalyst loadings yield high TONs
Note legali
Avvertenze
Danger
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 3 Oral - Aquatic Chronic 4 - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Articoli
QuinoxP*: Air-Stable and Highly Efficient and Productive Chiral Ligands
AEM3-944 | UAT | Prefill feature for related product categories not working
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.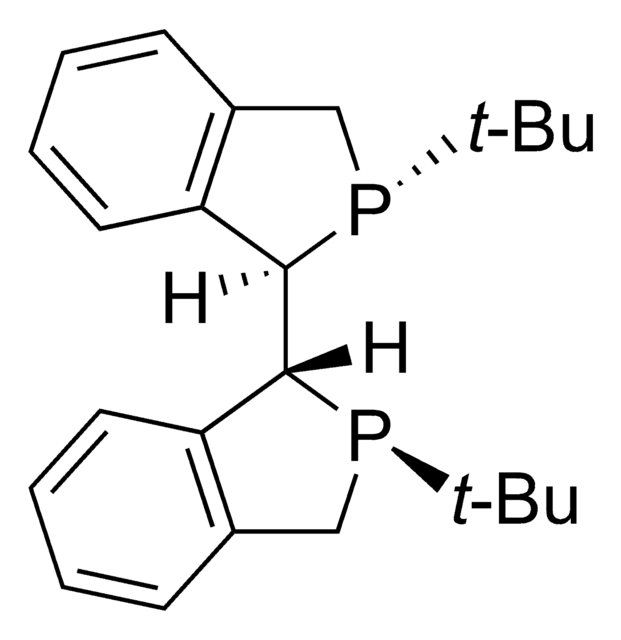
![(R)-1-[(SP)-2-(Diphenylphosphino)ferrocenyl]ethyldicyclohexylphosphine ≥97%](/deepweb/assets/sigmaaldrich/product/structures/245/493/2ae2dd8a-65cc-4aba-9a1f-1292eb1ad8e0/640/2ae2dd8a-65cc-4aba-9a1f-1292eb1ad8e0.png)
![(+)-1,2-Bis[(2S,5S)-2,5-dimethylphospholano]benzene kanata purity](/deepweb/assets/sigmaaldrich/product/structures/319/912/cec7b70f-bf7c-4a96-9f11-a73ae892e34c/640/cec7b70f-bf7c-4a96-9f11-a73ae892e34c.png)