317519
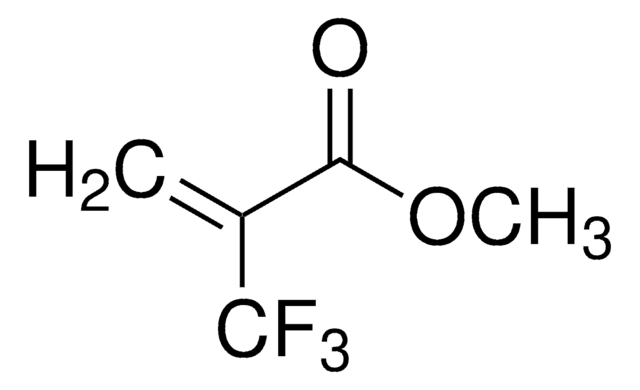
Methyl 2-acetamidoacrylate
98%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
H2C=C(NHCOCH3)CO2CH3
Numero CAS:
Peso molecolare:
143.14
Numero MDL:
Codice UNSPSC:
12162002
ID PubChem:
NACRES:
NA.23
Prodotti consigliati
Saggio
98%
P. ebollizione
104 °C/8 mmHg (lit.)
Punto di fusione
50-52 °C (lit.)
Stringa SMILE
COC(=O)C(=C)NC(C)=O
InChI
1S/C6H9NO3/c1-4(6(9)10-3)7-5(2)8/h1H2,2-3H3,(H,7,8)
SMWNFFKPVLVOQQ-UHFFFAOYSA-N
Categorie correlate
Descrizione generale
Conjugated addition of secondary amines, imidazole and pyrazole to methyl 2 methyl 2-acetamidoacrylate in the presence of a catalyst results in the formation of β-Dialkylamino-α-alanine and β-(N-heteroaryl)-α-alanine derivatives. Methyl-2-acetamidoacrylate (M2AA) is an anti-inflammatory agent. The catalytic reaction of methyl 2-acetamidoacrylate with Grignard′s reagents affords α-amino esters. M2AA can form thermosensitive copolymers with methyl acrylate.
Methyl ester of 2-acetamidoacrylate . methyl 2-acetamidoacrylate (Me-2-AA) is a di-unsaturated α-amino acid derivative. methyl-2-acetamidoacrylate exihibits anti -inflammatory properties, it is very effective against lipopolysaccharide (LPS)- induced nitric oxide production by RAW 264.
Applicazioni
Methyl 2-acetamidoacrylate can undergo [2+2] cycloaddition (Michael–Dieckmann-type reaction) with ketene diethyl acetal to yield the cyclobutane core. It may be used in rhodium-catalyzed 2-alkenylpyrrole formation.
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
235.4 °F - closed cup
Punto d’infiammabilità (°C)
113 °C - closed cup
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Thermosensitive properties of a novel poly(methyl 2-acetamidoacrylate-co-methyl acrylate)
Okamura H, et al.
European Polymer Journal, 38(4), 639-644 (2002)
Madeleine E Kieffer et al.
Journal of the American Chemical Society, 134(11), 5131-5137 (2012-03-07)
The tandem Friedel-Crafts conjugate addition/asymmetric protonation reaction between 2-substituted indoles and methyl 2-acetamidoacrylate is reported. The reaction is catalyzed by (R)-3,3'-dibromo-BINOL in the presence of stoichiometric SnCl(4), and is the first example of a tandem conjugate addition/asymmetric protonation reaction using
Joel A Tang et al.
Chemical communications (Cambridge, England), 47(3), 958-960 (2010-11-17)
A system that provides a sustained hyperpolarized (1)H NMR signal in an aqueous medium is reported. The enhanced signal lasts much longer than typical (1)H T(1) values, uncovering new possibilities for implementing hyperpolarized (1)H NMR/MRI experiments or performing kinetics studies
Synthesis of ?-amino acid derivatives by copper(I)-catalyzed conjugate addition of grignard reagents to methyl 2-acetamidoacrylate,
Cardellicchio C, et al.
Tetrahedron, 26(36), 4387-4390 (1985)
The Vinyl Moiety as a Handle for Regiocontrol in the Preparation of Unsymmetrical 2, 3?Aliphatic?Substituted Indoles and Pyrroles.
Huestis MP, et al.
Angewandte Chemie (International Edition in English), 123(6), 1374-1377 (2011)
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.